Sigma-2 Receptor Ligand Binding Modulates Association between TSPO and TMEM97
By Bashar M. Thejer, Vittoria Infantino, Anna Santarsiero, Ilaria Pappalardo, Francesca S. Abatematteo, Sarah Teakel, Ashleigh Van Oosterum, Robert H. Mach, Nunzio Denora, Byung Chul Lee, Nicoletta Resta, Rosanna Bagnulo, Mauro Niso, Marialessandra Contino, Bianca Montsch, Petra Heffeter, Carmen Abate, and Michael A. Cahill
Excerpt from the article published in the International Journal of Molecular Sciences. 24(7):6381. 28 March 2023. DOI: https://doi.org/10.3390/ijms24076381
Editor’s Highlights
- A close physical colocalization of Sigma-2 receptor (TMEM97/S2R) and the translocator protein (TSPO) was found in MIA PaCa-2 (MP) pancreatic carcinoma cells.
- The progesterone receptor membrane component 1 (PGRMC1), which has already been reported as a TMEM97/S2R interactor, co-immunoprecipitates with TSPO in MP cells but not in MCF7 breast adenocarcinoma cells.
- The different interacting partners in the two cell lines which we employed suggest that the TMEM97 interactome is context-dependent.
- The PGRMC1-TSPO-TMEM complex in MP cells is sensitive to the presence of S2R/TMEM97 endogenous ligand 20S-OHC, as well as exogenous ligands, suggesting is involved in S2R cell biology.
Abstract
Sigma-2 receptor (S2R) is a S2R ligand-binding site historically associated with reportedly 21.5 kDa proteins that have been linked to several diseases, such as cancer, Alzheimer’s disease, and schizophrenia. The S2R is highly expressed in various tumors, where it correlates with the proliferative status of the malignant cells. Recently, S2R was reported to be the transmembrane protein TMEM97. Prior to that, we had been investigating the translocator protein (TSPO) as a potential 21.5 kDa S2R candidate protein with reported heme and sterol associations. Here, we investigate the contributions of TMEM97 and TSPO to S2R activity in MCF7 breast adenocarcinoma and MIA PaCa-2 (MP) pancreatic carcinoma cells. Additionally, the role of the reported S2R-interacting partner PGRMC1 was also elucidated. Proximity ligation assays and co-immunoprecipitation show a functional association between S2R and TSPO. Moreover, a close physical colocalization of TMEM97 and TSPO was found in MP cells. In MCF7 cells, co-immunoprecipitation only occurred with TMEM97 but not with PGRMC1, which was further confirmed by confocal microscopy experiments. Treatment with the TMEM97 ligand 20-(S)-hydroxycholesterol reduced co-immunoprecipitation of both TMEM97 and PGRMC1 in immune pellets of immunoprecipitated TSPO in MP cells. To the best of our knowledge, this is the first suggestion of a (functional) interaction between TSPO and TMEM97 that can be affected by S2R ligands.
1. Introduction
The sigma-2 receptor (S2R) is an 18–21 kDa membrane protein that is a potential biomarker of the proliferative status of solid tumors [1,2,3]. Many S2R ligands modulate disease states [3,4,5] and can induce cell death [6], leading to an interest in their therapeutic potential [1,3,7]. Although the sigma-1 receptor (S1R) was successfully cloned in 1996 [8] and crystallized in 2016 [9], cloning and structural characterization of S2R has proved more elusive. Major progress was made when a photoactivated S2R ligand became cross-linked to PGRMC1, identifying a PGRMC1-containing complex with S2R activity [10]. This led to considerable controversy [11,12,13,14,15,16] over the non-identity of PGRMC1 with S2R after some papers referred to PGRMC1 as “PGRMC1/S2R”, assuming that PGRMC1 was S2R (e.g., [17,18,19]).
In 2017, the transmembrane protein 97 (TMEM97), which is also known as meningioma-associated protein 30 (MAC30), was identified as a gene coding for S2R activity in tumor cell lines [20]. Attenuation of TMEM97 by siRNA in PC-12 cells resulted in a proportional reduction of S2R ligand binding. Although overexpression of TMEM97 in Sf9 insect cells caused an increase of saturable [3H]DTG binding, the same was not true of PGRMC1 [20]. Mach and colleagues apparently resolved the controversy of how TMEM97 could be the S2R [20] by showing that TMEM97 forms a complex with PGRMC1 that directs enhanced internalization of the low-density lipoprotein receptor (LDLR), thereby revealing that TMEM97 could participate in protein-protein interactions [21]. In 2021, separate ligand-bound crystal structures were solved for TMEM97 with two respective known ligands, which enabled the structural prediction of hundreds of novel S2R ligands: two of which were also solved as ligand-bound crystal structures [22].
Prior to the 2017 publication that S2R activity was associated with TMEM97 [20], we had been independently investigating the translocator protein (TSPO) as a potential 18–21 kDa candidate S2R protein. In a preliminary PGRMC1-HA co-immunoprecipitation (co-IP) and mass spectrometric protein identification assay, TSPO was identified as a potential PGRMC1-interacting protein among a list of many identified proteins, so we hypothesized that could be associated with S2R activity in MIA PaCa-2 (MP) pancreatic adenocarcinoma cells. Consequently, we hypothesized that TSPO expression could be associated with S2R activity in MP cells. TSPO, also known as the peripheral benzodiazepine receptor (PBR), is a mitochondrial transmembrane protein with reported heme- and cholesterol-binding activities [23], which is thought to be a critical cholesterol transporter [24]. The previous association of TSPO with steroidogenesis has been stringently criticized [25]. Once TMEM97 was identified as S2R, we hypothesized that TSPO could be its interactor, as shown for PGRMC1 [21]. Using the nanomolar-affinity (S2R Ki = 11 nM) and green-emitting fluorescent S2R ligand SW120 [10], we started our investigation on the possible interrelation between TMEM97 and TSPO in MP cells. The data obtained prompted us to evaluate the presence of TMEM97 and TSPO, in a panel of functionally different cell lines, by saturation analysis with the appropriate radioligands. In agreement with previous data reporting higher density of S2R in several lines of breast and pancreatic cancer cells than in the normal (immortalized) cells [26,27,28], pancreatic adenocarcinoma MP and breast adenocarcinoma MCF7 cells were selected because of the density of the TMEM97 and TSPO proteins. Thus, in MP and MCF7, we assessed the association possibility between TMEM97, PGRMC1, and TSPO by proximity ligation assay (PLA), co-IP, and fluorescent staining. Note that all the assays employed (PLA, co-immunofluorescence, or co-IP) can detect the presence of proteins that are bound in higher-order protein complexes. Therefore, none of them can prove direct physical contact between the two respective proteins, although PLA epitopes from two target proteins must be within 40 nm to obtain a signal [29].
In MCF7 cells, the association between TMEM97 and TSPO but not between TSPO and PGRMC1 was detected. In MP cells, which exhibited the highest abundance of TSPO, co-IP also demonstrated the association between PGRMC1 and TSPO. Importantly, 20-(S)-hydroxycholesterol (20S-OHC) has recently been proposed as an endogenous TMEM97 ligand [30]. Thus, we studied its effects, together with the impact of other small TMEM97-interacting molecules, on the TSPO-TMEM97 association. These data together suggest context/cell-dependent interactomes for TSPO and TMEM97 that can modulate S2R ligand-binding/activity.
2. Results
2.1. TSPO and TMEM97 Are Bound by the S2R Ligand SW120 in MP Cells
We preliminary investigated whether TSPO could influence the binding of S2R ligands to cells using the fluorescent S2R ligand SW120. We attenuated TMEM97 by siRNA or TSPO levels by stably transfecting MP cells with shRNA-expressing lentivirus and compared the reduction in SW120 binding in the TMEM97- or TSPO-attenuated cells (Figure 1). TMEM97 mRNA was reduced by approximately 80%, as assayed by RT-PCR (Figure 1A). This was accompanied by an approximately 20% reduction in the binding of SW120 to MP cells, as assayed by the detection of the median fluorescence intensity (MFI) of cells separated by flow cytometry (Figure 1B). Knockdown of TSPO mRNA in MP cells containing stably transduced lentiviral shRNA resulted in approximately 70% reduction in TSPO shRNA cells relative to scrambled shRNA (Scr shRNA) control cells (Figure 1C). The protein level knockdown of TSPO is shown in Figure S1C–E. This led to an approximately 50% reduction of SW120-dependent MFI (Figure 1D). SW120 binding to S1R in the MP cells did not significantly contribute to the SW120 fluorescence observed because cells were also incubated with 1.8 µM SKF10.047 to block S1R, compared to solvent DMSO as a control. This level of SKF10.047 is reported to block S1R so that S1R did not contribute to the SW120-generated signal in accordance with the S2R versus S1R selectivity of SW120 [31,32].
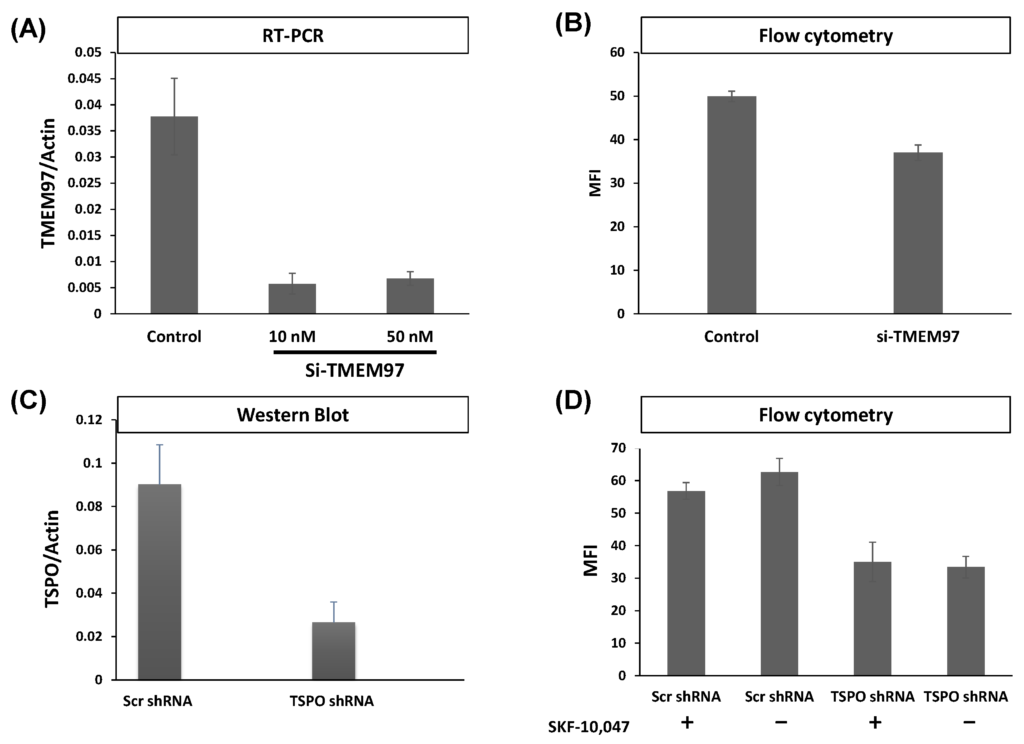
TMEM97 and TSPO attenuation both reduce S2R ligand-binding by SW120.
(A) Gene expression by RT-PCR showing the attenuation of TMEM97 mRNA in MP cell lines. The knockdown of TMEM97 by siRNA (either 10 or 50 nM per treatment) was confirmed by more than 80% (t-test p < 0.001). (B) Flow cytometry assessment of SW120 binding to MP cells (n = 6). Median fluorescence intensity (MFI) was determined by measuring the FITC signal (FL1). Kaluza software was used to analyze the data. Attenuation of TMEM97 (10 nM siRNA) resulted in significant SW120 reduction (t-test p < 0.0001). (C) Detection of TSPO expression levels by Western blot after TSPO shRNA knockdown compared with scramble shRNA control. The image of the Western blot is supplied in Figure S1E. Six independent clones were generated after transduction with viral particles, with all showing visible attenuation normalized to actin. A significant reduction of approximately 70% was achieved (t-test p < 0.00001). (D) The chart shows a flow cytometry evaluation of SW120 binding after TSPO knockdown by shRNA. MP cells were incubated with SKF-10,047 or DMSO as a negative control. All cells were incubated with 30 nM of SW120 in complete media for 30 min at 37 °C. Treatment with SKF-10.047 had no significant impact on SW120 binding (Two-way ANOVA). In contrast, attenuation of TSPO resulted in a significant reduction in the SW120 signal (Two-way ANOVA p < 0.0001) in both conditions. Six replicates of each cell line were tested.
2.2. TSPO and TMEM97 Affect the Same SW120-Binding Sites in MP Cells
In the above results, SW120 binding was influenced by levels of both TSPO and TMEM97. This could be due to the same molecule binding two different binding sites at each protein, in which case, we would expect to see additive effects upon simultaneous attenuation. Alternatively, both proteins could be affecting the same binding sites, in which case, the effects of a double knockout (DKO) should be about the same as each individual knockout. To better understand the effect posed by both proteins, we generated DKO MP cells using shRNA attenuation for TSPO and transiently transfected siRNA for TMEM97. The attenuation of each protein caused approximately equal reductions in SW120 binding. Strikingly, the DKO cells showed no additive reduction in signal strength (Figure 2, left-hand bars). The lack of an additive effect on SW120 binding upon simultaneous attenuation of both TSPO and TMEM97 suggests that TSPO and TMEM97 levels affect the same S2R ligand-binding sites.
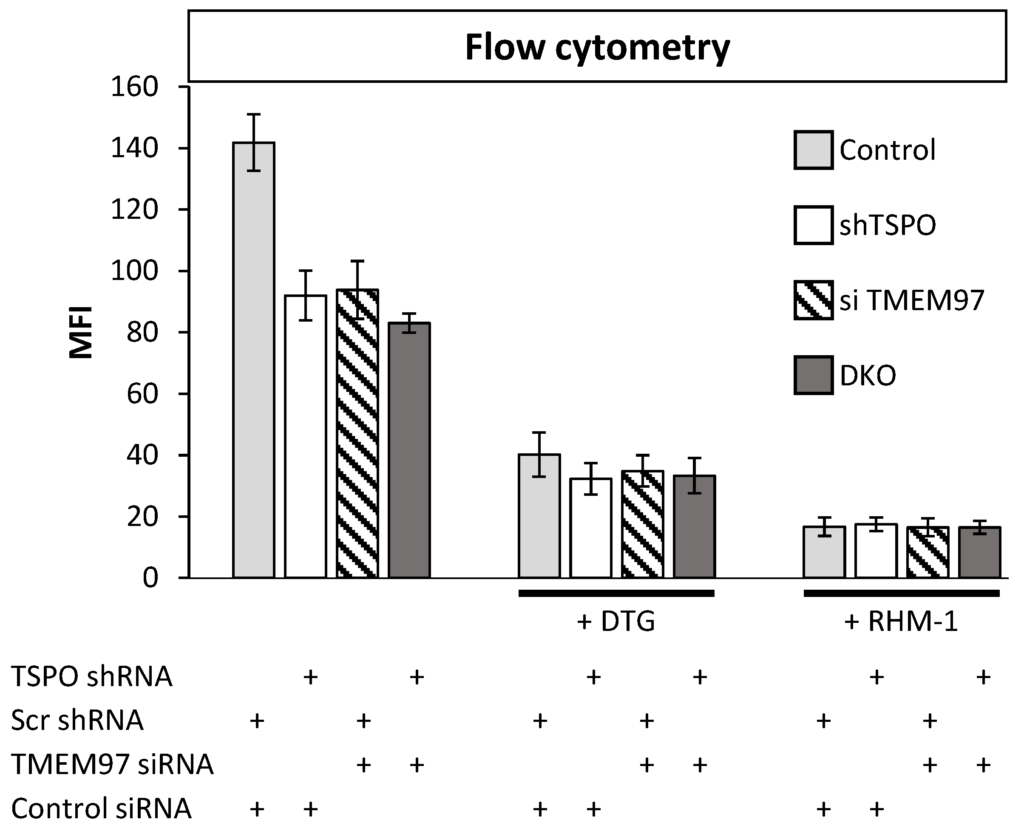
Double knockout (DKO) of TMEM97 and TSPO produced no further reduced SW120 signal in MP cells.
The figure shows the first incubating condition for four differently treated cell lines. That includes Scr control cell lines, cells with an attenuated level of TSPO or TMEM97, and cells with the attenuated level of both TSPO and TMEM97 (DKO). Those cells were incubated with SW120 for 30 min at 37 °C. All treatments were significantly different from control cells (ANOVA, post hoc Tukey’s HSD, p < 1 × 10−7). The right bars show the same condition, but with the treatment of DTG or RHM-1 prior to incubation with SW120. Cells were incubated with complete media supplemented with 10 nM DTG or 4 nM of RHM-1 for 1 h at 37 °C. For DTG-treated cells (+DTG), the DTG-treated control cells were significantly different at the p < 0.0001 level from the untreated control cells. All DTG-treatment conditions (shTSPO, si TMEM97, and DKO) were significantly different from DTG-treated control at the p < 0.005 level (t-test on + DTG cells only). All RHM1-treated cells (+ RHM-1) were significantly different from respective untreated cells at the p < 0.001 level (t-test on + RHM-1 cells only). There was no significant difference between Control, shTSPO, siTMEM97, or DKO for RHM-1 and DTG-treated cells (t-test on + DTG or + RHM-1 treated cells).
DTG exhibits binding to both S1R and S2R, whereas the S2R antagonist RHM-1 has 300-fold specificity for S2R relative to S1R and can be used in binding competition assays to discriminate between S1R and S2R binding [33,34]. Upon addition of either DTG or RHM-1, all the cell lines exhibited a significant reduction in SW120 binding (Figure 2), indicating that the SW120 binding to MP cells, as measured by flow cytometry MFI, was at least mostly due to the S2R. Following the rationale of Weng et al. [34], who argue that RHM-1 is a superior selective ligand for S2R over DTG, these results indicate that the SW120 fluorescence observed by flow cytometry MFI values in Figure 2 can be attributed to specific SW120-S2R complexes. While the residual signal after RHM-1 treatment may be due to the unspecific binding of SW120, taken together, these results suggest that TSPO and TMEM97 contribute cooperatively to SW120 binding to the S2R. This suggests that either the binding site is formed upon the interaction between these two proteins or that the close proximity between the two proteins may allosterically stabilize the S2R binding site. Further research will be required to characterize this situation.
In control experiments, we confirmed that all respective antibodies gave positive immune fluorescence signals, which were attenuated by the knockdown of PGRMC1 or TSPO, respectively (Figure S1).
2.3. TSPO Colocalizes with TMEM97 in MP Cells
The results from the SW120 experiments prompted us to perform proximity ligation assays in MP cells to evaluate the close proximity of the proteins. Thus, we evaluated the following interactions: PGRMC1 + TMEM97, PGRMC1 + TSPO, and TSPO + TMEM97. We obtained positive PLA signals for the TMEM97/TSPO antibody pair (Figure 3). These results are consistent with TSPO and TMEM97 forming a protein complex, which is possibly required for S2R activity in these cells. We observed low levels of PLA signal between PGRMC1 and TMEM97. Differences to the higher levels observed by Riad et al. in HeLa cells [21] may reflect cell-specific differences. Riad et al. also used a polyclonal anti-PGRMC1 antibody, whereas we employed an anti-HA tag antibody against the HA tag of exogenously expressed PGRMC1-HA. It is additionally conceivable that the TMEM97/PGRMC1 interaction is sensitive to the presence of the HA tag at the PGRMC1 C-terminus. No positive PLA signals for the PGRMC1/TSPO antibody pair were found. However, taken together, our results show a clear and previously undescribed interaction between TSPO and TMEM97. Since the TMEM97-PGRMC1 interrelation has been previously established and could be cell type-specific, but the TSPO-TMEM97 interaction was novel, we concentrated further efforts on the latter.
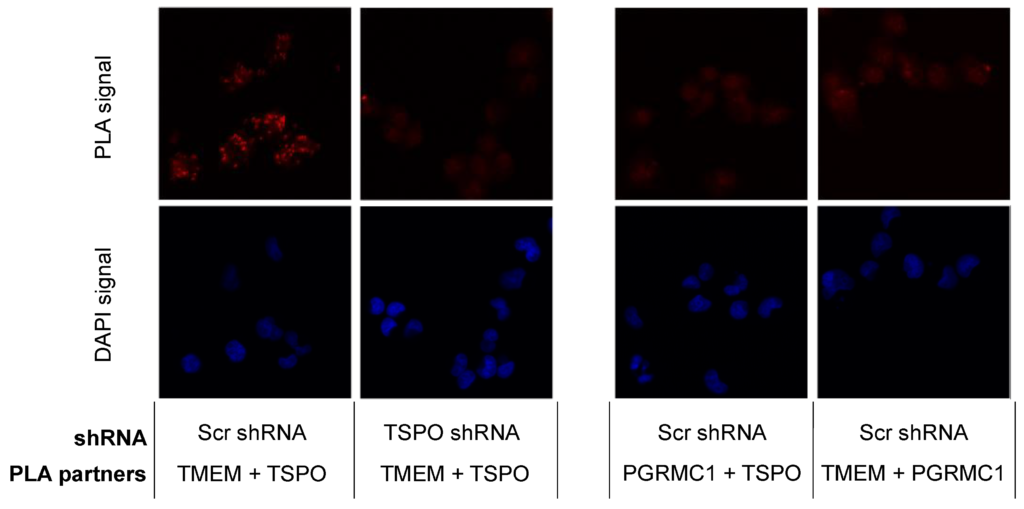
TSPO is colocalized with SW120 receptor S2R in MP cells.
The left panels represent PLA results for TMEM97 (TMEM) and TSPO pairing. The positive signal was observed compared with the negative signal after attenuating TSPO. The right panels show two negative PLA signals for TMEM97 + PGRMC1 and PGRMC1 + TSPO. Those negative observations were similar to a negative control without primary antibodies. Red corresponds to the signal amplified by the Duolink PLA reaction described in Section 4.12. Blue represents DAPI staining of DNA. Each discrete blue patch represents one cell nucleus (imaged at 20× magnification).
2.4. Saturation Analyses by Radioligand Binding to Detect TMEM97 and TSPO in Functionally Different Cell Lines
Four cell lines were then selected to identify the most appropriate cell models for the protein-protein TSPO-TMEM97 interaction study, which may be context-dependent. Rat glioma C6 cells are routinely used in TSPO-binding experiments [35,36,37] and express S2R [38]. By contrast, colorectal adenocarcinoma LoVo cells were previously reported to present S2R with a low density [28]. MP and MCF7 cells both display S2R, with the latter cell being employed as a model to study S2R-mediated activity [39,40,41,42]. We also generated an MCF7 subline (shMCF7TMEM97), which exhibited lower TMEM97 protein levels due to shRNA knockdown (see paragraph below). As depicted in Table 1 and Figure S2, saturation analysis detected the presence of receptors for TSPO ligands in all studied cell lines to different extents. The highest amount was detected in MP cells (Bmax = 7.3 pmol/mg of protein) and the lowest in MCF7 cells (Bmax = 1.02 pmol/mg of protein).
| Bmax, pmol/mg of Protein | Bmax, pmol/mg of Protein | |
|---|---|---|
| Cell Lines | TSPO | TMEM97/S2R |
| C6 rat glioma | 3.60 | 2.15 |
| MCF7 | 1.02 | 1.795 |
| shMCF7TMEM97 | 2.06 | 0.891 |
| LoVo | 3.43 | 0.54 |
| Mia PaCa-2 (MP) | 7.30 | 1.14 |
Analogously, S2R ligand binding was observed in all the selected cells except for LoVo. The highest level was measured in MCF7 and C6 cells (Bmax = 2.02 and 2.15 pmol/mg of protein, respectively) (Table 1, Figure S2). It is noteworthy that receptors for ligands binding TSPO and S2R are expressed in the same cell lines (except for LoVo cells) and that the reduction in S2R ligand-binding observed in shMCF7TMEM97 cells (Bmax = 0.891 pmol/mg of protein) is accompanied by an increase in TSPO ligand-binding (Bmax = 2.06 pmol/mg of protein) compared with the MCF7 parental line, suggesting a compensatory effect that is consistent with a hypothesized functional link existing between the expression patterns of TMEM97 and TSPO.
2.5. Generation of MCF7 and MP Cells with Attenuated Levels of TMEM97/S2R
With the aim of generating models to study the protein-protein interaction in more detail in the two selected cell lines, MCF7 and MP cells, we attenuated the level of TMEM97. In MP cells, TMEM97 levels were reduced by siRNA, as described by Alon et al. [20], and the level of gene expression was measured by RT-PCR. In both 10 nM and 50 nM siRNA concentrations, TMEM97 mRNA was significantly reduced by around 80% (t-test p < 0.0001) (Figure 1A), leading to significantly reduced SW120 binding (t-test p < 0.0001) (Figure 1B). Here, we also included an S1R-specific competitive control, SKF10.047, to ensure the S2R specificity of SW120 binding.
In MCF7 cells, TMEM97 levels were reduced by shRNA, and the amount of residual TMEM97 was measured by saturating [3H]DTG (Table 1, Bmax = 0.891 pmol/mg protein in shMCF7TMEM97 cells vs. Bmax = 1.795 pmol/mg protein in WT cells; Figure S2H,D, respectively). As recently reported, a corresponding significant reduction in the binding of two structurally different green-emitting S2R-specific fluorescent ligands (i.e., F412 and NO1) was detected [43].
2.6. TMEM97 Co-Immunoprecipitates with TSPO
In order to support the potential protein-protein associations between TSPO and TMEM97 and/or PGRMC1, we performed co-IP experiments with a specific anti-TSPO primary antibody. In MCF7 cells, we found that TMEM97 was detected in immune pellets obtained by precipitation of TSPO but that PGRMC1 was not (Figure 4A,B). The co-immunoprecipitation data in MCF7 cells were consistent with confocal microscopy results in the same cells, where colocalization was observed between a fluorescent S2R ligand and TSPO (Figure S3A) but not with PGRMC1, which exhibited a nuclear localization (Figure S3B). TSPO and PGRMC1 were immunostained by specific antibodies against each protein, while S2R (as a surrogate for TMEM97) was stained via a recently reported CY5-labeled S2R-specific ligand [43].
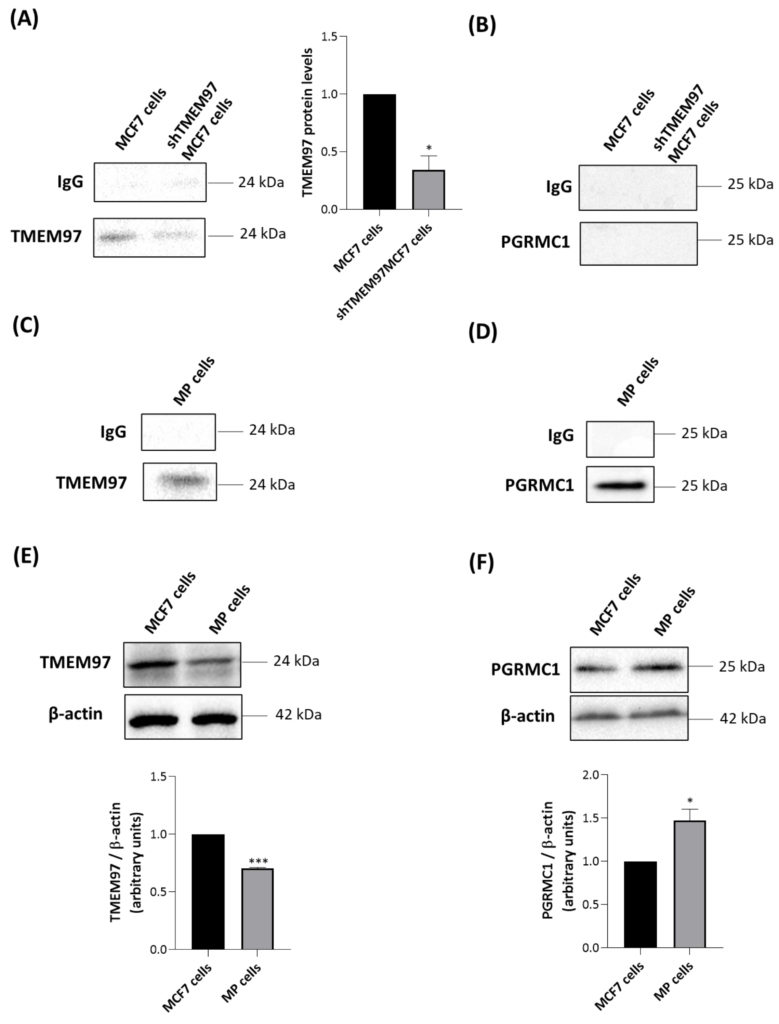
TSPO co-immunoprecipitates only with TMEM97 in MCF7 cells and with both TMEM97 and PGRMC1 in MP cells.
MCF7 and shTMEM97MCF7 cells were immunoprecipitated with an antibody directed against TSPO and analyzed by Western blot with anti-TMEM97 (A) or anti-PGRMC1 (B) antibodies. TMEM97 protein levels in shTMEM97MCF7 cells were normalized versus proteins in MCF7 cells. MP cells were immunoprecipitated with an antibody directed to TSPO and analyzed by Western blot with anti-TMEM97 (C) or anti-PGRMC1 (D) antibodies. IgG is the negative control. Western blot analyses were performed to evaluate the protein levels of TMEM97 (E) and PGRMC1 (F) in MCF7 MP cells. In (E,F) protein levels were normalized against β-actin. Western blotting data are representative of at least three independent experiments. Graph bars were used to represent the means ±SD (error bars). Statistical significance of the differences was evaluated by using Student’s t-test (* p < 0.05, *** p < 0.001).
As expected, the intensity of a TMEM97-related band was greater in parental MCF7 than in shTMEM97MCF7 cells (Figure 4A). In both MCF7-based cell lines, no signal was found when immune complexes precipitated with anti-TSPO were blotted with anti-PGRMC1 antibody (Figure 4B). By contrast, in MP cells, we observed that both TMEM97 (Figure 4C) and PGRMC1 (Figure 4D) co-immunoprecipitated with TSPO. This suggests the existence of a possible multiprotein complex formed by TMEM97, TSPO, and PGRMC1 in MP cells. Alternatively, TSPO could be present in separate protein complexes containing either PGRMC1 or TMEM97, despite the absence of a positive PGRMC1/TSPO PLA signal.
To illuminate the difference observed between the two tumor cell lines, we performed Western blot experiments of whole cell lysates to evaluate the expression levels of TMEM97 and PGRMC1. TMEM97 was detected at higher levels in MCF7 than in MP cells (Figure 4E), confirming the saturation analyses performed with the S2R radioligand DTG (Table 1). PGRMC1 was detected at higher levels in MP than in MCF7 cells (Figure 4F). Therefore, more abundant expression of PGRMC1 in MP than in MCF7 cells could explain the detected association between TSPO and PGRMC1 in MP but not in MCF7 cells.
2.7. 20S-OHC Reduced the Associations TSPO-TMEM97 and TSPO-PGRMC1 in MP Cells
MP cells were treated for 24 h with 20S-OHC, which was recently proposed as an endogenous TMEM97 ligand [30], and immunoprecipitated with an anti-TSPO antibody. The analysis performed via Western blot revealed that 20S-OHC reduced the presence of both TMEM97 and PGRMC1 in TSPO immune precipitates (Figure 5). In fact, both TMEM97 (Figure 5A, upper panel) and PGRMC1 (Figure 5B, lower panel) protein bands were lower by more than half in 20S-OHC treated cells than in untreated control cells.
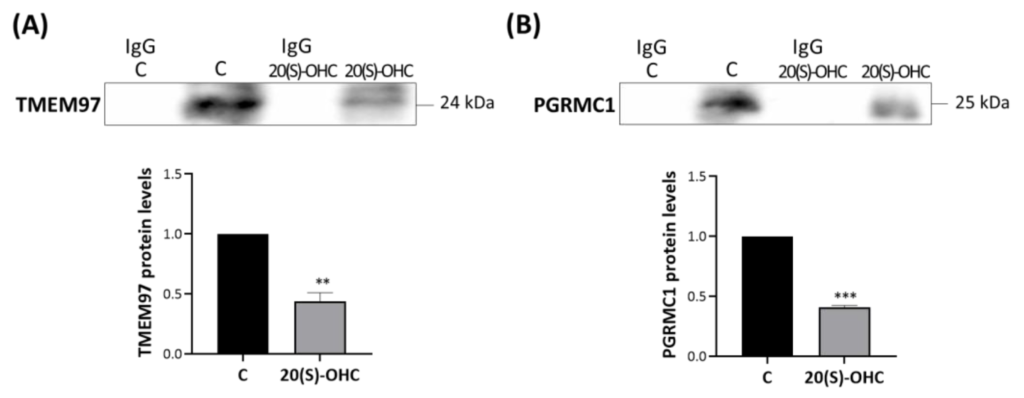
20S-OHC reduces levels of both TMEM97 and PGRMC1 co-immunoprecipitated with TSPO.
MP cells were treated for 24 h with 20S-OHC, immunoprecipitated with an antibody directed against TSPO, and the immune pellets were analyzed by Western blot with anti-TMEM97 (A) or anti-PGRMC1 (B) specific antibodies. IgG is the negative control for the anti-TSPO experimental treatment immunoprecipitation. Western blotting data are representative of at least three independent experiments. TMEM97 (A) and PGRMC1 (B) protein levels in 20S-OHC treated cells were normalized versus proteins in untreated cells (C) and reported in the respective graph bars as means ± SD (error bars). Statistical significance of the differences was evaluated by using Student’s t-test (** p < 0.01, *** p < 0.001).
2.8. Effect of Siramesine Oxalate Salt and FA10 Hydrochloric Salt on TSPO Interactions-With TMEM97 and PGRMC1 in MP Cells
We thus investigated the role of synthetic TMEM97/S2R ligands on these interactions. MP cells were treated for 2 h with 10 μM siramesine oxalate salt (S) [44] or FA10 ([1-(3-(6,7-dimethoxy-3,4-dihydroisoquinolin-2(1H)-yl)propyl]-5-methoxy-1,2,3,4-tetrahydroquinoline hydrochloric salt) [45], immunoprecipitated with an anti-TSPO antibody and analyzed via Western blotting with specific antibodies directed against TMEM97 (Figure 6A) and PGRMC1 (Figure 6B). Notably, the former compound is a reference S2R agonist, whereas the latter is a novel, highly selective S2R ligand devoid of antiproliferative activity (in contrast to siramesine). Siramesine oxalate salt significantly increased the interactions between TSPO-TMEM97 (Figure 6A) and TSPO-PGRMC1 (Figure 6B) in comparison with untreated MP cells. In the presence of FA10 hydrochloric salt, the interaction between TSPO and PGRMC1 was stronger than in control cells (Figure 6B), but no significant changes were observed with respect to the TSPO-TMEM97 interaction (Figure 6A).
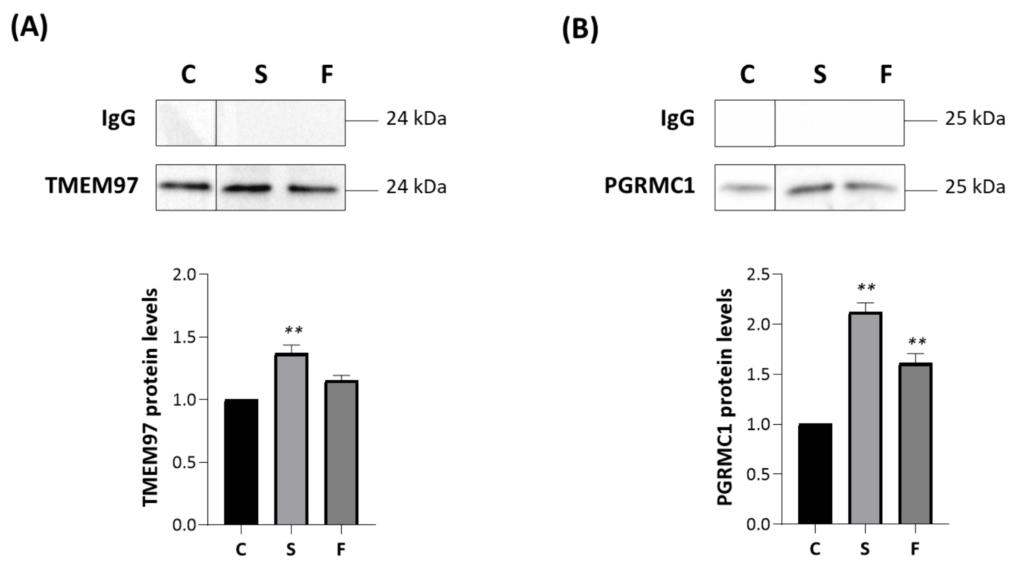
Siramesine oxalate salt and FA10 hydrochloric salt affect the associations TSPO-TMEM97 and TSPO-PGRMC1.
MP cells were treated for 2 h with siramesine oxalate salt (S) and FA10 hydrochloric salt (F), immunoprecipitated with an antibody directed against TSPO and analyzed by Western blot with anti-TMEM97 (A) or anti-PGRMC1 (B) specific antibodies. IgG is the negative control for the TSPO antibody. Western blotting graphs depict the results of at least three independent experiments. TMEM97 and PGRMC1 protein levels in treated cells were normalized versus proteins in untreated cells (C) and reported in the respective graph bars (lower panels) as means ± SD (error bars). Statistical significance of the differences was evaluated by using one-way ANOVA followed by Dunnett’s multiple comparisons test (** p < 0.01).
3. Discussion
For many years S2R was known as an enigmatic S2R ligand-binding site, which was attributed to protein species of 21.5 kDa [3]. Before the report that TMEM97 binds S2R ligands [20], we had reasoned that TSPO (predicted mass 18.8 kDa) could be post-translationally modified to possibly generate the 21.5 kDa species, which is why we were investigating TSPO. After Alon et al. [20] reported that TMEM97 binds to S2R ligands, we attempted to reconcile our results with theirs. As shown above, we observed proximal and functional interactions between TMEM97 and TSPO, as well as with the previously reported TMEM97 interaction partner PGRMC1 [21].
TMEMs are upregulated in cancers, where they are associated with tumor progression, invasion, and metastasis [46]. Also known as MAC30, TMEM97, is an endoplasmic reticulum-resident membrane protein [20]. TMEM97 was identified as a cholesterol-regulating gene by functional RNAi screening [47]. We show that both TSPO and TMEM97 are required for SW120 binding. Importantly, attenuation of each protein in the double knockdown did not cause an additive reduction of the SW120 signal, which would be expected if SW120 independently binds the two proteins. These results, suggesting that these proteins form a complex, prompted further investigations that we conducted in two cell line models selected for their TMEM97/S2R and TSPO expression patterns, i.e., MCF7 and MP cells. Due to the previously detected interaction between TMEM97 and PGRMC1 in protein complexes, also PGRMC1 was included in the investigation. Results from PLA and co-IP experiments support an interaction between TMEM97 and TSPO in both cell lines. In co-IP pellets of TSPO immunoprecipitates, no PGRMC1 was detected in MCF7 cells, in agreement with the lack of colocalization of the two proteins, as detected by confocal microscopy. On the other hand, PGRMC1 was detected in co-IP pellets of TSPO immunoprecipitates in MP cells, in contrast to the PLA results in MP cells, where PGRMC1-TSPO interaction was not detected. Taken together, these results suggest that the interaction of TSPO and PGRMC1 does not correspond to a close proximity revealable by PLA. Alternatively, an inappropriate orientation could hamper the annealing of the primers to generate a PLA signal, or TSPO interaction may block the antigen for the PGRMC1 antibody or vice versa. Notably, the differences observed between the two cells for co-IP partners suggest a context-dependent interactome that may also reflect the density of the proteins.
Overall, all the performed experiments together suggest protein-protein interactions that involve different partners in different cells: TSPO + TMEM97 in MCF7 and TSPO + TMEM97 and TSPO + PGRMC1 in MP cells. These interactions appear to be functional. The administration of endogenous (20S-OHC) or exogenous (siramesine and FA10) TMEM97/S2R ligands affects the co-IP profile, suggesting still unexplored pathways for S2R ligands-mediated activity.
The nature of the TMEM97-TSPO association remains unclear. We do not know whether the two proteins are in a direct physical complex or are merely closely juxtaposed in a common protein complex. The presence of a positive PLA signal argues for at least intimate proximity.
In HeLa cells, it has been demonstrated that PGRMC1 and TMEM97 are present in the complex bound by S2R ligands because a photoactivatable S2R ligand bound to the S2R formed a covalent cross-link with PGRMC1 upon photo-activation [10], and the two proteins colocalize in a protein complex as demonstrated [21,48]. Furthermore, we know that the TMEM97-binding site for S2R ligands is situated within the lipid bilayer in cells [22]. Finally, recognized TSPO ligands are pharmacologically distinct from S2R ligands [49].
Given these premises, we reason that TSPO may allosterically facilitate S2R ligand binding. This could be under a model where the TSPO-TMEM97 complex facilitates a TMEM97 conformation amenable to PGRMC1 binding (in MP cells) and/or to S2R ligand binding, thereby facilitating a change in TMEM97-complex status. In either model, TSPO could either remain present as part of the TMEM97-containing complex or be dissociated by allosteric protein conformational changes upon altered TMEM97-complex status. In the latter scenario, the dissociated TMEM97-containing component and/or the TSPO-containing component could be free to perform subsequent downstream functions (signal propagation) that were unavailable to the intact TSPO-TMEM97 complex (analogously to the ability of Gα and Gβγ subunits to perform downstream functions after their dissociation following ligand engagement by G-protein-coupled receptors [50]). However, note that Figure 5 suggests that TSPO may dissociate from both TMEM97 and PGRMC1 upon binding of the endogenous 20S-OHC S2R ligand. All the above scenarios remain hypothetical and will require subsequent experimental investigation.
TSPO is primarily reported as a mitochondrial protein [51], although it has been reported in the nucleus of spermatogenic cells [52] and in urinary exosomes [53], which implies that occupancy of the post-endosomal cytoplasmic vesicle compartment is at least sometimes possible. Future studies will need to address the subcellular location of the TMEM97/TSPO complex reported in this present study.
It is notable that all three proteins under consideration (TMEM97, TSPO, and PGRMC1) have interrelated biological attributes. TSPO is involved with steroid and heme biology [49] (which is the reason we began examining it), as is PGRMC1 [21,48,54,55], whereas TMEM97 is involved in regulating cholesterol levels [47,56], and its endogenous ligand appears to be 20S-OHC [30].
PGRMC1 is the archetypal member of the membrane-associated progesterone receptor (MAPR) family. This eukaryotic family forms a distinct sub-class of the cytochrome b5 domain superfamily, characterized by tyrosinate-mediated heme chelation, as opposed to the His-2 heme chelation of classical cytochrome b5 proteins [57,58]. Intriguingly, a group of bacterial proteins from the candidate phyla radiation (CPR) bacteria shares tyrosinate heme chelation with MAPR proteins. These CPR bacterial proteins have been named cytb5MY proteins (MAPR-like cytochrome b5 with tyrosine/Y-heme chelation). It is unclear whether eukaryotic MAPR proteins originated from a CPR bacterial cytb5MY protein or whether the cytb5MY proteins arose by horizontal gene transfer of an MAPR gene into a CPR bacterium [58]. However, in the context of a putative biological relationship between mammalian PGRMC1 and TSPO, it is intriguing that the reconstructed ancestral CPR bacterial operon containing a cytb5MY protein appears to have also contained a TSPO gene, as well as two other cytochrome b5 proteins and a putative ferric-reductase enzyme. The operon also contained a two-component inducible element, and all proteins possessed at least one trans-membrane helix, possibly suggesting that the operon encoded a redox-related inducible membrane complex [58]. All of those proteins have heme prosthetic groups [58,59] if we include the heme-associated TSPO [12]. While the CPR bacterial TSPO genes are less resemblant to eukaryotic TSPOs than other prokaryotic TSPOs, and therefore eukaryotic TSPO is not of CPR bacterial origin, it is possible that the colocation of TSPO and an MAPR-like cytb5MYprotein in CPR bacterial operons is reflective of a functional relationship between these two protein classes, possibly related to heme biology and redox functions. This remains highly conjectural yet provides hypotheses that may usefully direct future research.
…
5. Conclusions
In conclusion, our reported co-immunoprecipitation and colocalization of TMEM97/S2R with TSPO is a novel finding whose characterization requires further investigation. In addition, PGRMC1, which has already been reported as a TMEM97/S2R interactor, co-immunoprecipitates with TSPO in MP cells but not in MCF7. The different interacting partners in the two cell lines which we employed suggest that the TMEM97 interactome is context-dependent. Importantly, the PGRMC1-TSPO-TMEM complex in MP cells is sensitive to the presence of S2R/TMEM97 endogenous ligand 20S-OHC, as well as exogenous ligands, suggesting that the complex is involved in S2R cell biology. Overall, the previously unreported protein-protein interactions of these three proteins shed new light on their biology and functions and deserve further investigation that may disclose unexplored mechanisms of action and therapeutic potentials.