The Sigma-2 Receptor Selective Agonist Siramesine (Lu 28-179) Decreases Cocaine-Reinforced Pavlovian Learning and Alters Glutamatergic and Dopaminergic Input to the Striatum
By Anna M. Klawonn, Anna Nilsson, Carl F. Rådberg, Sarah H. Lindström, Mia Ericson, Björn Granseth, David Engblom, and Michael Fritz
Excerpt from the article published in Frontiers in Pharmacology, 10 October 2017, Sec. Experimental Pharmacology and Drug Discovery, Volume 8 – 2017, DOI: https://doi.org/10.3389/fphar.2017.00714
Editor’s Highlights
- The sigma-2 receptor agonist Siramesine already has been tested in a clinical-phase II trial for anxiety.
- Siramesine significantly attenuated acquisition, expression, and drug-primed reinstatement of cocaine-induced place preference. Siramesine did this most effectively at a dose of 0.3 mg/kg.
- σ2Rs provide negative control over glutamate release from cortical structures in the nucleus accumbens (NAc).
- sigma-2 receptors regulate neurocircuitry responsible for positive reinforcement and thereby play a role in cocaine-reinforced Pavlovian behaviors.
Abstract
Drug addiction is a chronic, debilitating disease that affects millions of people around the world causing a substantial societal burden. Despite decades of research efforts, treatment possibilities remain limited and relapse represents the most treatment-resistant element. Neurosteroid sigma-1 receptors have been meticulously studied in psychostimulant reinforced Pavlovian learning, while the sigma-2 receptor subtype has remained unexplored. Recent development of selective sigma-2 receptor ligands have now made it possible to investigate if the sigma-2 receptor system is a potential target to treat drug addiction. We examined the effect of the sigma-2 receptor agonist Siramesine (Lu 28-179) on cocaine-associated locomotion, Pavlovian learning, and reward neurocircuitry using electrophysiology recordings and in vivo microdialysis. We found that Siramesine significantly attenuated conditioned place preference acquisition and expression, as well as it completely blocked cocaine-primed reinstatement. Siramesine, in a similar manner as the selective sigma-1 receptor antagonist BD 1063, decreased acute locomotor responses to cocaine. Immunohistochemistry suggests co-expression of progesterone receptor membrane component 1/sigma-2 receptors and vesicular glutamate transporter 1 in presynaptic boutons of the nucleus accumbens (NAc). Whole-cell voltage clamp recordings of neurons in the NAc indicated that Siramesine decreases the presynaptic release probability of glutamate. Further, we demonstrated, via in vivo microdialysis, that Siramesine significantly decreased cocaine-evoked dopamine release in the striatum of freely moving mice. Collectively, these findings demonstrate that sigma-2 receptors regulate neurocircuitry responsible for positive reinforcement and thereby play a role in cocaine-reinforced Pavlovian behaviors.
Introduction
Drug addiction is a chronic relapsing disorder with detrimental effects for the affected individuals and, despite decades of scientific effort, treatment possibilities remain scarce and relapse rates high (Volkow et al., 2016). Therefore, exploring new treatment targets against relapse is pivotal. Neurosteroid sigma-receptors (σRs) have previously been suggested to play a role in addiction pathology. Two subtypes of the σR have been identified based on drug selectivity and molecular mass: σ1R and σ2Rs (Cobos et al., 2008). Whereas the role of σ1Rs in addiction has been studied, very little is known about that of σ2Rs.
Cloning of the σ1R (Hanner et al., 1996) led to advances in the understanding of its function and linked it to modulation of various neurotransmitters (e.g., glutamate, GABA, serotonin, acetylcholine, etc.) and several neuropathologies (Cobos et al., 2008). In contrast, the identity of the σ2R remains controversial. A recent study identified the σ2R as the progesterone receptor membrane component 1 (PGRMC1) (Xu et al., 2011), and a follow-up study showed that the PGRMC1 protein levels correlate with the binding activity of a σ2R-selective probe (SW120) in the rat brain (Zeng et al., 2016). These findings, however, are debatable due to newer studies showing that PGRMC1 knockdown/out or knock-in interventions do not change σ2R binding of [3H]-DTG (1,3-di-o-tolylguanidine; a shared σ1R and σ2R ligand) in affinity assays (Abate et al., 2015; Chu et al., 2015). Nonetheless, compounds with selective binding affinities for σ2Rs relative to σ1Rs have been developed (Guo and Zhen, 2015). H. Lundbeck A/S generated the most selective ligand currently available: Lu 28-179/Siramesine. This is a highly selective σ2R agonist (σ2R Ki = 0.12 nM; σ1R 17 nM) (Perregaard et al., 1995) initially developed as an anxiolytic agent (Sánchez et al., 1997) and later found to have antidepressant-effects in rats (Sánchez and Papp, 2000). Furthermore, Siramesine has been used for labeling σ2R binding sites in rats, demonstrating the presence of σ2Rs in the nucleus accumbens (NAc), caudate putamen (CPu), and ventral tegmental area (VTA) (Søby et al., 2002). More recently, other σ2R ligands have been explored in cocaine addiction-related behaviors. 5-Bromo-N-[4-(6,7-dimethoxy-3,4-dihydro-1H-isoquinolin-2-yl)-butyl)]-2,3-dimethoxy-benzamide was found to antagonize cocaine-induced hyperlocomotion in mice (Lever et al., 2014) and Katz et al. (2016)observed effects of σ2R antagonists (CM398, CM353) on cocaine primed DTG self-administration. Finally, σ2R antagonist SN79 was shown to attenuate DTG-induced dopamine levels in the striatum (Garcés-Ramírez et al., 2011). Interestingly, the possible effects of Siramesine have not yet been explored in cocaine-reinforced behaviors.
In order to address the role of σ2Rs in behaviors relevant to psychostimulant addiction we tested Siramesine in acquisition, expression, and drug-primed reinstatement of cocaine-induced conditioned place preference (CPP), as well as in acute cocaine-induced hyperlocomotion using mice. Since Siramesine diminished some of the cocaine reinforced behaviors, we decided to employ a comparative pharmacological approach using the σ1R selective antagonist, BD 1063 (McCracken et al., 1999; Liu et al., 2005; Sabino et al., 2009). Furthermore, we elucidated how Siramesine affects glutamatergic input to cells in the NAc via whole-cell slice recordings and cocaine-evoked dopamine release within the striatum (NAc and CPu) via in vivo microdialysis in freely moving mice.
…
Results
Locomotor Measurements
To assess whether BD 1063, a σ1R selective antagonist, or Siramesine, a σ2R selective agonist, have an effect on baseline locomotor activity, mice were injected with saline or a high or a low dose of either compound. No differences in locomotor activity were found between the saline and the BD 1063 groups, Figure 1A [two-way ANOVA F(2,37) = 1.273, p > 0.05]. Whereas Siramesine at a dose of 0.3 mg/kg significantly decreased baseline locomotor activity after 5, 20, and 25 min [two-way ANOVA F(2,37) = 3.593, p < 0.05], Figure 1B. Next, we evaluated if BD 1063 or Siramesine were able to attenuate cocaine-induced hyper-locomotion. Mice received either saline, BD 1063 (3 or 10 mg/kg) or Siramesine (0.3 or 1 mg/kg) 10 or 60 min before an acute 15 mg/kg cocaine injection. Subsequently, the animals were monitored for 30 min. BD 1063 significantly decreased acute cocaine-induced hyper-locomotion during the last 15 min of the recorded behavior, Figure 1C [two-way ANOVA F(2,37) = 8.599, p < 0.01]. Treatment with Siramesine reduced the overall acute locomotor-effect of cocaine, Figure 1D [two-way ANOVA F(2,37) = 4.09, p < 0.05].
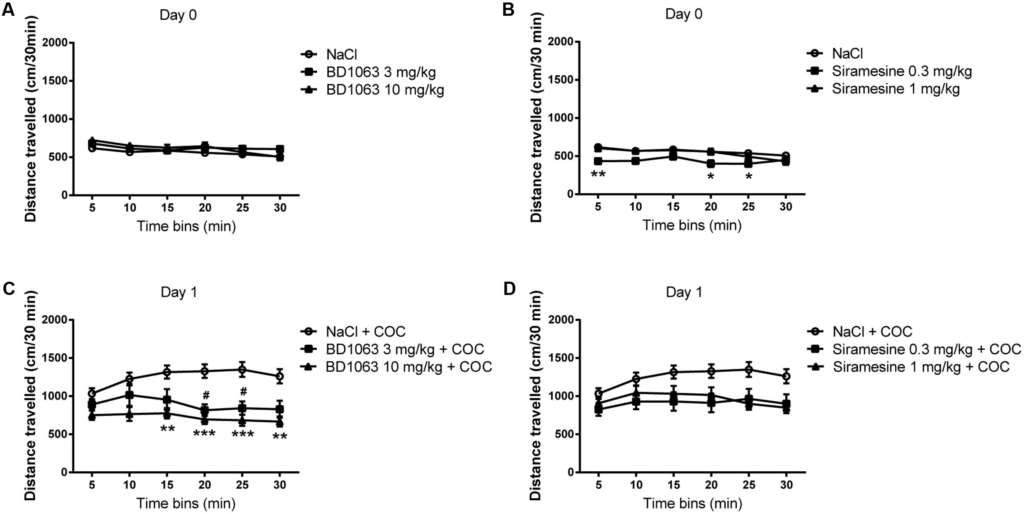
Locomotor Measurements.
Mean ± SEM of baseline locomotor measurements during exposure to (A) NaCl (n = 24) vs BD 1063 [3 mg/kg (n = 8) or 10 mg/kg (n = 8)] and (B) NaCl (n= 24) vs Siramesine [0.3 mg/kg (n = 8) or 1 mg/kg (n = 8)]. BD 1063 had no effect on baseline locomotor activity, whereas Siramesine significantly attenuated it at 5, 20, and 25 min. (C)BD 1063 mitigates acute cocaine-induced hyperlocomotion in the last half of the 30-min tracking interval. (D) Siramesine also attenuated the acute cocaine-induced increase in locomotor activity as designated by significance in the two-way ANOVA. Locomotor measurements from NaCl-treated control mice were pooled and represented in both graphs. #,∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001. Two-way ANOVA followed by Bonferroni’s post hoc test.
Acquisition of Cocaine-Associated Place Preference
To elucidate if σ2Rs are involved in acquiring cocaine-associated Pavlovian learning in the same manner as σ1Rs, we used a place preference paradigm. Mice received saline or BD 1063 (1, 3, or 10 mg/kg) 10 min before each cocaine training session. As expected, 1 and 10 mg/kg BD 1063 significantly attenuated the acquisition of cocaine place preference, Figure 2A [one-way ANOVA F(3,23) = 7.245, p < 0.01]. In a second experiment, we administered Siramesine (0.1, 0.3, or 1 mg/kg) 60 min prior to the cocaine training sessions. All doses of Siramesine attenuated the acquisition of cocaine place preference, Figure 2B [one-way ANOVA F(3,23) = 13.66, p < 0.01]. To ensure that neither BD 1063 nor Siramesine were inherently rewarding or aversive, we used an effective dose of each compound (10 and 0.3 mg/kg, respectively) as an unconditioned stimulus. Neither substances caused avoidance or preference behavior, Figure 2C.
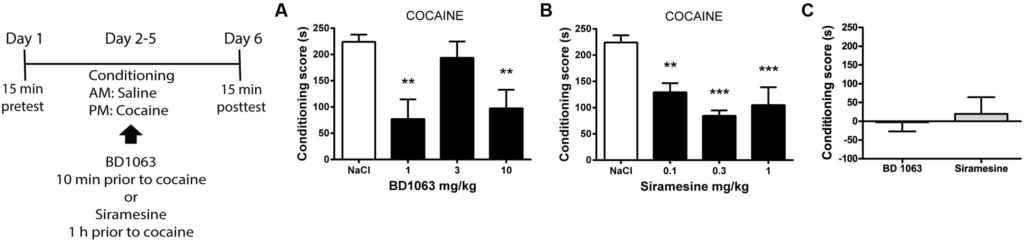
Acquisition of cocaine-associated place preference.
Mean ± SEM of (A) the conditioning score for cocaine-induced place preference with or without BD 1063 [NaCl control (n = 10) vs 1 mg/kg (n = 5) vs 3 mg/kg (n = 6) vs 10 mg/kg (n = 6)]. (B) Siramesine attenuates the acquisition of cocaine-induced place preference [NaCl control (n = 10), 0.1 mg/kg (n = 6), 0.3 mg/kg (n = 6), 1 mg/kg (n = 5)]. CPP scores from NaCl-treated control mice were pooled and represented in both graphs. (C) Neither 10 mg/kg BD 1063 (n = 6) nor 0.3 mg/kg Siramesine (n = 6) cause an avoidance or approach behavior. ∗∗p < 0.01, ∗∗∗p < 0.001. One-way ANOVA followed by Dunnett’s post hoc test.
Expression of Cocaine-Induced Place Preference
We next investigated if σ2Rs play a role similar to σ1Rs during expression of learned cocaine associations. To do so, mice that had undergone cocaine-conditioning, were given either saline or BD 1063 (1, 3, or 10 mg/kg) 10 min prior to the place preference test. All three doses of BD 1063 significantly lowered the expression of cocaine-place preference, Figure 3A [one-way ANOVA, F(3,37) = 4.247, p < 0.05]. In a subsequent experiment, conditioned mice were administered either saline or Siramesine (0.1, 0.3, or 1 mg/kg) 60 min before the posttest. Siramesine was also able to significantly reduce the expression of cocaine-induced place preference at a dose of 0.3 mg/kg, Figure 3B [one-way ANOVA, F(3,35) = 5.374, p < 0.01]. Subsequently, we examined if the effects of BD 1063 and Siramesine were specific to cocaine-reinforced motivational learning or affect natural reward learning as well. Whereas BD 1063 did not alter Nutella-induced place preference, Siramesine significantly attenuated the expression of place preference to this palatable food, Figure 3C [one-way ANOVA, F(2,12) = 12.66, p < 0.01]. To assess, if this effect was due to metabolic changes, we monitored regular food intake for 12 h in animals injected with either saline or Siramesine (0.3 or 1 mg/kg). No effect of Siramesine on food-intake was found, Figure 3D [two-way ANOVA, F(2,45) = 3.567, p > 0.05].
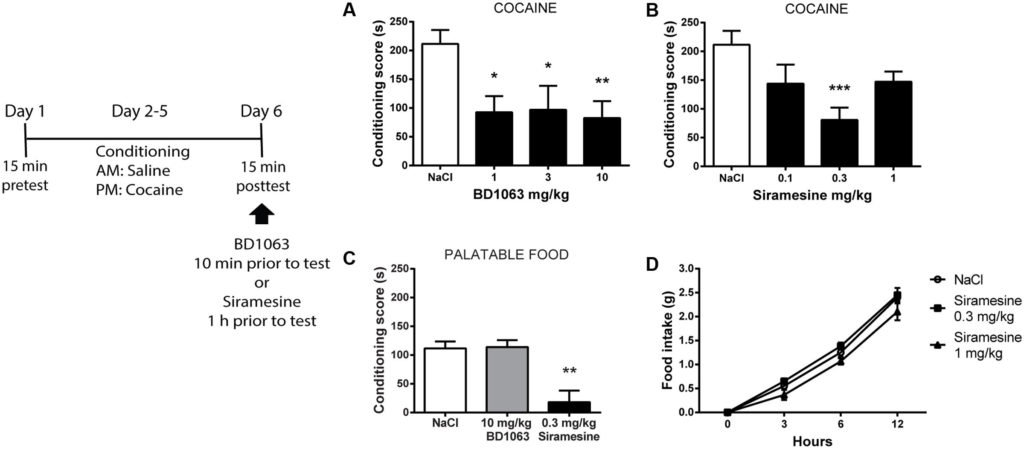
Expression of cocaine-induced place preference.
Both, BD 1063 (A) (n = 12, 7, 9, and 13) and Siramesine (B) (n = 12, 8, 10, and 9) attenuate the expression of cocaine place preference as represented with mean ± SEM. CPP scores from NaCl-treated control mice were pooled and represented in both graphs. (C) Siramesine, but not BD 1063, reduces the expression of palatable food place preference (n = 5, 5, and 5). (D)Siramesine has no effect on cumulative food intake (n = 6, 6, and 6). ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001. One-way ANOVA followed by Dunnett’s post hoc test.
Drug-Primed Reinstatement of Cocaine Place Preference
Finally, we tested if BD 1063 or Siramesine are capable of preventing drug-primed reinstatement of cocaine-induced place preference. Mice were trained to express cocaine-CPP and subsequently underwent an extinction protocol. Animals that displayed significant loss of preference were submitted to reinstatement the following day. Mice received saline, BD 1063 (1, 3, or 10 mg/kg) or Siramesine (0.1, 0.3, or 1 mg/kg), 10 or 60 min, respectively, prior to reinstatement using a low dose of cocaine (5 mg/kg). The mice were then immediately tested for re-expression of cocaine-seeking behavior. BD 1063 was ineffective at attenuating cocaine-primed reinstatement, Figure 4A [one-way ANOVA, F(3,45) = 0.8225, p > 0.05]. In contrast, drug-primed reinstatement of cocaine seeking was abolished in mice that had received at least 0.3 mg/kg of Siramesine, Figure 4B [one-way ANOVA, F(3,36) = 7.261, p < 0.01].
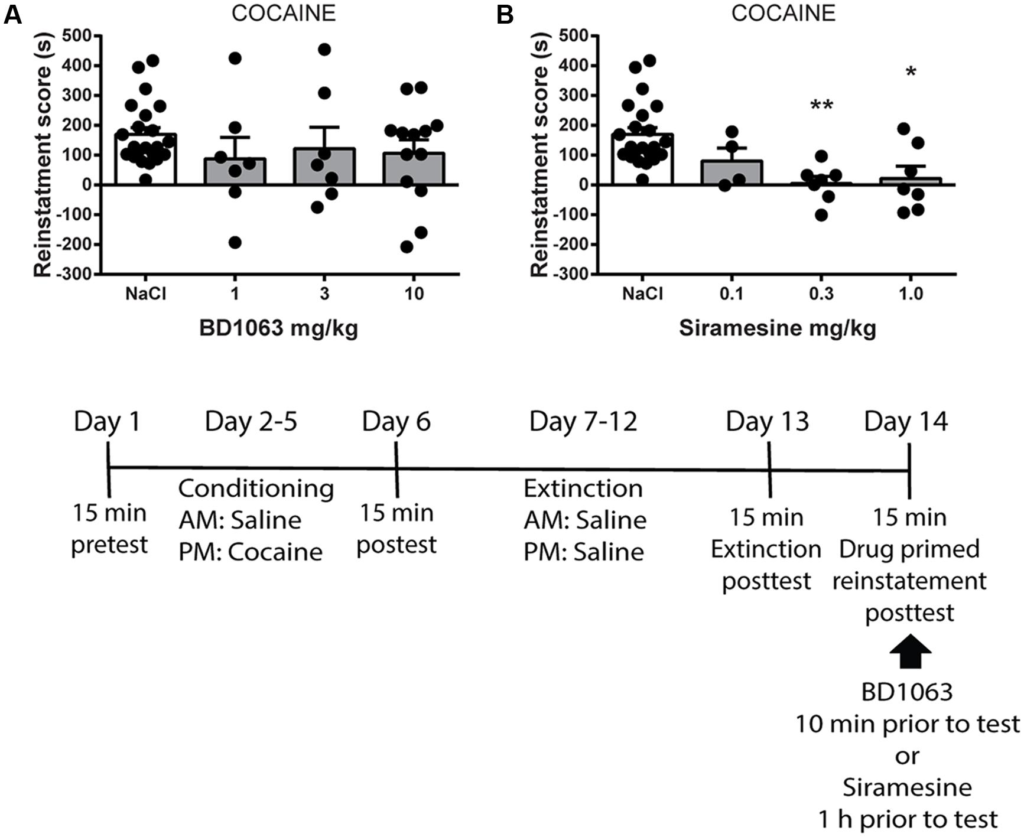
Drug-primed reinstatement of cocaine-place preference.
(A) BD 1063 does not prevent drug-primed reinstatement (n = 22, 7, 7, and 13) as shown by mean ± SEM. In contrast, (B)Siramesine blocks cocaine-primed reinstatement of a previously extinguished place preference (n = 22, 4, 7, and 7). CPP scores from NaCl-treated control mice were pooled and represented in both graphs. ∗p < 0.05, ∗∗p < 0.01. One-way ANOVA followed by Dunnett’s post hoc test.
Whole-Cell Patch Clamp Recordings of Nucleus Accumbens Neurons
Medial prefrontal cortex (mPFC) glutamatergic synaptic transmission to NAc neurons is necessary for reinstatement of drug seeking (LaLumiere and Kalivas, 2008) and has been recognized as a primary target for cocaine (Suska et al., 2012). σ2R agonism has previously been shown to inhibit voltage-gated calcium channels, including the P- and N-types that are important for triggering transmitter release, indicating that σ2Rs could modulate the efficiency of synaptic vesicle release (Zhang and Cuevas, 2002). To determine if the putative σ2R (PGRMC1) is present on glutamatergic terminals in the NAc, we performed fluorescence immunohistochemistry. Presynaptic terminals of glutamatergic neurons were identified by VGLUT1 immunoreactivity. PGRMC1-labeling was found at approximately 40% of such synapses in the NAc, as illustrated by co-localization between PGRMC1 and VGLUT1 immunoreactivity, Figures 5A,B. It should be noted that despite the eminent expression of PGRMC1 on VGLUT1-positive terminals, several PGRMC1 puncta in the NAc were VGLUT1-negative. To investigate if σ2Rs have an effect on synaptic vesicular release, we performed whole-cell voltage-clamp recordings of NAc neurons. Siramesine significantly reduced the frequency (Student’s paired t-test, p < 0.05) of spontaneous excitatory synaptic events (sEPSCs), without affecting their amplitude, Figures 5C–E. The glutamatergic identity of these events was confirmed at the end of recording sessions by glutamate receptor antagonists DNQX and APV, Figure 5C. An acute decrease in sEPSC frequency is commonly interpreted as a decrease in transmitter release probability from the presynaptic bouton.
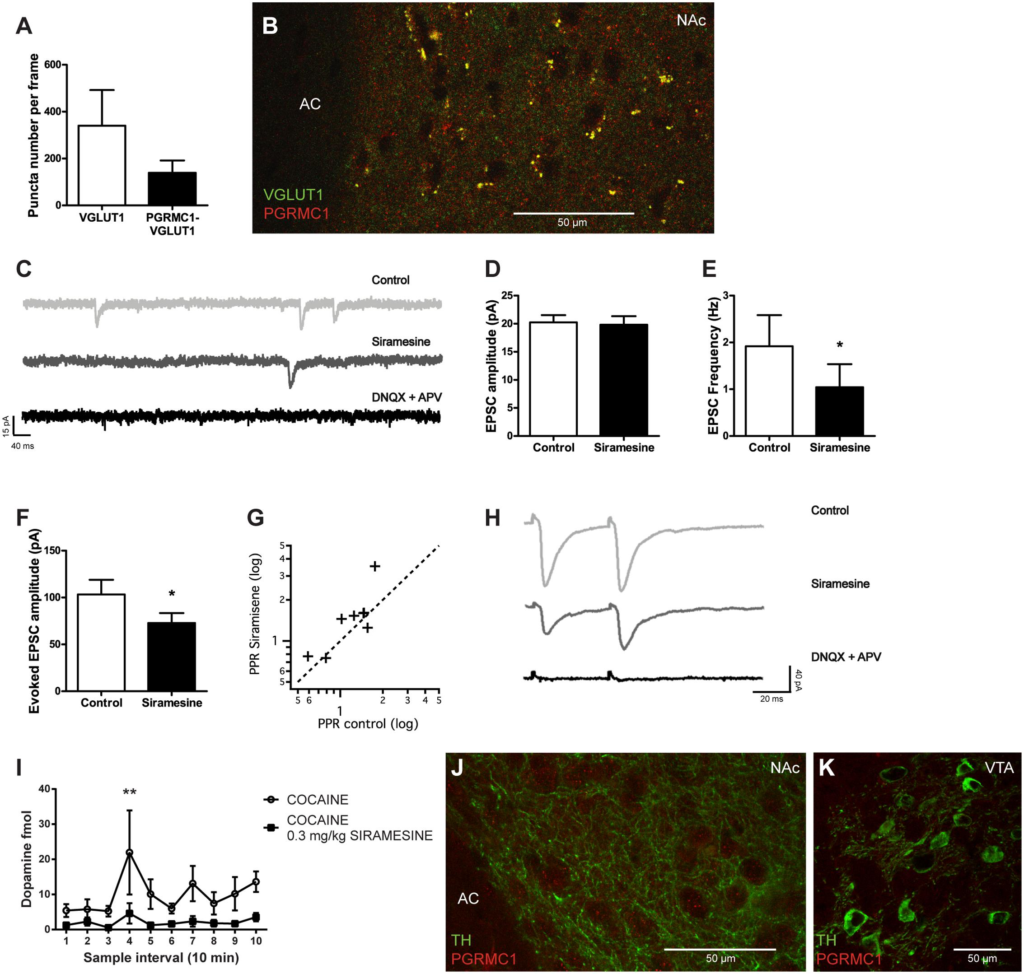
Effects of Siramesine on glutamate release probability in the nucleus accumbens and dopamine input to the striatum.
Mean ± SEM of (A) numbers of VGLUT1 expressing boutons vs VGLUT1 boutons co-localized with PGRMC1/σ2R puncta in the NAc (n = 4). (B) A 63× confocal image showing co-localization of VGLUT1 (green) and PGRMC1/σ2R (red) in terminals adjacent to cell somas of NAc near the anterior commissure (AC). (C) Spontaneous EPSCs before and after bath-application of drugs. Siramesine decreases the frequency of spontaneous events, while DNQX and APV abolishes them completely. (D)Siramesine does not affect the amplitude of spontaneous EPSCs (E) but significantly decreases their frequency as represented in mean ± SEM (n= 8). (F) Siramesine decreases the amplitude of the first evoked EPSCs (n = 8), (G) and increases the paired pulse ratio (PPR) in six out of eight cells. (H) Example traces from the paired pulse stimulus, showing how Siramesine reduces EPSC amplitude and increases PPR. Evoked EPSCs are abolished by DNQX and APV. (I) Dopamine concentrations in the striatum (NAc and CPu) prior to and following cocaine administration. When Siramesine was injected 30 min prior to sampling, it significantly decreased cocaine-induced dopamine levels in the striatum. Cocaine was i.p. injected at the third sample-interval (NaCl n = 8, Siramesine n = 9). (J) A 63× confocal image of TH-positive fibers (green) and PGRMC1/ σ2R (red) in terminals adjacent to cell somas of NAc near the anterior commissure (AC). (K) A 63× confocal image of VTA TH-positive neurons (green) without any PGRMC1/σ2R (red) co-labeling of immunoreactivity. ∗p < 0.05, ∗∗p < 0.01. Student’s paired t-test or two-way ANOVA followed by Bonferroni’s post hoc test.
To further investigate the effect of Siramesine on transmitter release, cortico-striatal fibers were stimulated with a tungsten electrode. Evoked EPSCs recorded in NAc neurons were significantly smaller after application of the σ2R agonist, Figures 5F,H (Student’s paired t-test, p < 0.05). Further, during paired pulse stimulation (50 ms separation), the paired pulse ratio (EPSC2/EPSC1) was increased in six out of eight cells, Figures 5G,H. These findings support that the activation of σ2Rs indeed reduces the efficiency by which synaptic vesicles are released at cortico-striatal synapses in the NAc. Hence, Siramesine could mediate its effect on positive reinforcement behavior by influencing the release-probability of neurotransmitters signaling reward.
In Vivo Microdialysis
To further investigate the role of σ2Rs in the reward neurocircuitry, we examined if Siramesine affects dopamine levels in the striatum using in vivo microdialysis. Mice were given either saline or 0.3 mg/kg Siramesine and were subsequently attached to the microdialysis tubing and placed in the infusion cage. After an initial 30 min period, which was used to stabilize flow and neurotransmitter exchange, three 10 min baseline samples were taken. Immediately after the third sample was taken, all mice received a 15 mg/kg cocaine injection and another seven samples were collected. Two-way ANOVA [F(9,150) = 1.723; p = 0.08 group-factor; F(1,150) = 26.11 p < 0.01 sample-factor] and post hoc analysis (Bonferroni multiple comparison; p < 0.01 for sample interval 4) revealed that Siramesine attenuated cocaine-evoked dopamine release in the striatum, Figure 5I. To determine if the protein PGRMC1 (the putative σ2R) is present on dopaminergic terminals in the striatum or on dopamine neurons in the VTA, we performed fluorescence immunohistochemistry targeting TH, the rate-limiting enzyme in catecholamine synthesis. We found no expression of PGRMC1 on TH-positive terminals in the striatum (example shown from NAc), Figure 5J, nor on TH-positive neurons in the VTA, Figure 5K.
Discussion
In this study, we used a preclinical model of Pavlovian reinforcement learning to demonstrate the potential of utilizing σ2Rs as treatment targets against psychostimulant drug addiction. The selective σ2R agonist Siramesine significantly attenuated acquisition, expression, and drug-primed reinstatement of cocaine-induced place preference. Siramesine did this most effectively at a dose of 0.3 mg/kg. It did so in a manner similar to the selective σ1R antagonists BD 1063 and previously reported BD 1047 (Maurice et al., 2002; Maurice and Romieu, 2004), with the exception that it proved to be more effective against drug-primed reinstatement than BD 1063. The two receptor subtypes also differed with respect to baseline locomotor activity, whereas σ1R antagonism had no effect, Siramesine significantly decreased locomotor movement. In our study and in others, BD 1063 robustly and reproducibly attenuated cocaine-induced hyperlocomotion (McCracken et al., 1999; Matsumoto et al., 2001), the same was observed for Siramesine. This is in accordance with recently published findings on a selective σ2R ligand (Lever et al., 2014).
We cannot exclude that the dissimilarities in effects between BD 1063 and Siramesine could be due to diverse pharmacokinetic properties, as the drugs were administered at different time-points and the specific pharmacokinetic profile of BD 1063 has not yet been fully characterized. But since we see effects of BD 1063 on cocaine-induced locomotion, acquisition, and expression of CPP, it seems unlikely that the compound would not be functionally active within this timeframe. Furthermore, similar studies on drugs of abuse-induced behaviors have reported effects after pre-treatment with BD 1063 within the same time-frame as our experiments (McCracken et al., 1999; Sabino et al., 2009). Hence, from our results, it seems likely that Siramesine possess certain different capabilities than BD1063 on natural (baseline locomotion and palatable food preference) and cocaine-related (drug-primed reinstatement) behaviors. This suggests that Siramesine, on the contrary to BD1063, may have a treatment potential against cocaine relapse.
A peculiar phenomenon of Siramesine is the absence of a clear dose–response relationship, as both high and low doses produced weaker effects in acquisition, expression, and reinstatement of CPP. This is not an uncommon occurrence for the σ-receptor system and has previously been reported in studies using σ1R agonists (review of literature, Cobos et al., 2008). The U-shaped dose–response relationship is explained by the biological process of hormesis. In the case of σ1R agonism, hormesis is assumed to occur as a consequence of the modulatory function these receptors exert over various neurotransmitter-systems (Cobos et al., 2008). Our findings indicate that similar mechanisms could be present during σ2R agonism. This is supported by Sánchez and Papp (2000), who found a comparable dose–response curve for Siramesine on depression-like phenotype. More specifically, σ1R hormesis has been proposed to occur as a consequence of postsynaptic adaptations via glutamatergic NMDA-receptors, but it could also be related to a presynaptic mechanism. Our findings would suggest the latter for the σ2R, since we see an effect on release-probability of glutamate.
Our results show that σ2Rs provide negative control over glutamate release from cortical structures in the NAc. It has been demonstrated that glutamatergic input to the NAc, including afferents from mPFC, drives reward seeking (Britt et al., 2012). The Siramesine-induced reduction in release probability at glutamatergic synapses with NAc neurons may explain the decrease in motivational drive toward cocaine and palatable food, which was observed in our study. Changes in excitatory input to medium spiny neurons (MSNs) of the NAc could elucidate the observed attenuation in cocaine evoked dopamine-release. It has previously been shown that a significant number of MSNs, projecting to the VTA, target GABAergic interneurons and thereby regulate dopamine tone (Xia et al., 2011). A decrease in excitability of such MSNs, due to Siramesine acting on glutamatergic terminals, could result in lowered dopamine release.
Furthermore, despite that PGRMC1 is widely expressed throughout the brain as shown with in situ hybridization (Allen Brain Atlas4), we observed no co-labeling with TH-positive neurons in the VTA nor on TH-positive terminals in the striatum. If the σ2R indeed is the same protein as PGRMC1, it is unlikely that the decrease in cocaine evoked dopamine levels is due to a direct effect of Siramesine on VTA dopamine neurons. Finally, we noticed the presence of a number of PGRMC1-positive puncta in the NAc that are VGLUT1- and TH-negative. These could as well contribute to the observed effects on behavior. Irrespectively, it is clear that Siramesine has significant effects on classical motivational neurocircuitry.
It is interesting to note, that whereas previous studies have shown a role for σ1R antagonism in treating drug addiction (McCracken et al., 1999; Maurice et al., 2002; Maurice and Romieu, 2004; Romieu et al., 2004; Sabino et al., 2009; Fritz et al., 2011), the effects we observed are due to σ2R agonism. This would indicate that the two specific σR-subtypes have dissimilar molecular responses to agonists and antagonists, or alternatively execute their function via opposing neurocircuitry. Some evidence support the first alternative. We found that σ2Rs decrease glutamate release in NAc. In contrast, it has been reported that σ1Rs enhance glutamate release in various structures (i.e., hippocampus and cortex) (Meyer et al., 2002; Dong et al., 2006). Similarly, our results showed σ2R agonism decreased dopamine release in the striatum, while a study using the selective σ1R agonist, PRE-084, demonstrated significantly increased dopamine levels (Garcés-Ramírez et al., 2011).
Yet, some dichotomies presented in the current literature on σ2Rs remain to be solved. A recent study (Katz et al., 2016) showed that cocaine primed self-administration of DTG (a combined σ1R and σ2R agonist) can be attenuated in a dose-dependent manner using σ2R selective antagonists (CM398 and CM353). This would indicate that σ2R agonism has positive reinforcing properties, which was not observed in our study. Further enquiries are needed to provide a better understanding of the identity and molecular mechanisms of σ2R signaling. Also, future preclinical investigations of Siramesine would be of importance in order to assess how it influences operant self-administration in rodents.
Finally, since Siramesine already has been tested in a clinical-phase II trial for anxiety, it would be relevant to expand clinical trials to test its efficacy in preventing relapse in patients being treated for cocaine addiction.

