ZC3H4 promotes pulmonary fibrosis via an ER stress-related positive feedback loop
By Hammad Ghafoor, Han Chu, Jie Huang, Mengling Chen, Sha Wang, Jing Wang, and Jie Chao
Excerpt from the article published in Toxicology and Applied Pharmacology, Volume 435, 2022, 115856, ISSN 0041-008X, https://doi.org/10.1016/j.taap.2021.115856
Editor’s Highlights
- ZC3H4 directly interacts with the Sigmar1 protein in the pulmonary fibroblast activation.
- Silicosis is a type of pulmonary fibrosis caused by exposure to dust particles containing a high level of free silica-containing silicon dioxide (SiO2).
- Fibrosis in pneumoconiosis and Idiopathic pulmonary fibrosis (IPF) is a chronic pathological process in which Zinc finger CCCH-type signaling molecules and the transforming growth factor-beta (TGF-β) pathway are involved.
- SiO2 exposure enhanced the MAPK and P13K/Akt phosphorylation pathways with the involvement of ZC3H4, a CCCH-type zinc finger protein.
- ZC3H4 induces ER stress with the participation of a positive feedback loop.
- ZC3H4 and Sigmar1 might act as novel therapeutic targets for pulmonary fibrosis.
Autors’ Highlights
- Increased ZC3H4 in pulmonary fibroblast was associated with fibroblast activation.
- ZC3H4 activated fibroblasts via the sigmar1/ER stress pathway.
- ER stress-regulated ZC3H4 via positive feedback.
- ZC3H4 maybe a novel therapeutic strategy for treating silicosis.
Abstract
Background: Pulmonary fibrosis is a sequela of many pulmonary diseases, such as pneumoconiosis and idiopathic pulmonary fibrosis. The principal characteristics of pulmonary fibrosis comprise myofibroblast proliferation, alveolar damage and deposition of extracellular matrix components, which cause abnormal lung structure remodeling and an irreversible decline in lung function; however, the detailed mechanisms remain unclear. The current study focused on the role of ZC3H4, a new member of the zinc finger protein family, in SiO2-induced pulmonary fibrosis.
Methods: The expression of ZC3H4 and fibroblast activation markers (COL1A1, COL3A1 and ACTA1) was measured by western blotting and immunofluorescence staining after SiO2 exposure (50 μg/cm2). The functional change in fibroblasts was studied with a scratch assay and a 3D migration assay. The CRISPR/Cas9 system was used to explore the regulatory mechanisms of ZC3H4 in pulmonary fibroblast cells.
Results: The expression levels of ZC3H4 and sigmar1 (a key regulator of ER stress) were increased in pulmonary fibroblast cells and were associated with fibroblast activation, as indicated by the increase in COL1A1, COL3A1 and ACTA1, as well as the migration ability. SiO2-enhanced fibroblast activation was attenuated by specific knockdown of ZC3H4 and inhibition of ER stress, demonstrating that ZC3H4 activated fibroblasts via the sigmar1/ER stress pathway. Interestingly, ER stress blockade also inhibited ZC3H4 expression, indicating the positive feedback regulatory mechanism of ER stress on ZC3H4.
Conclusions: Our results demonstrate that ZC3H4 and sigmar1 might act as novel therapeutic targets for silicosis, providing a reference for further pulmonary fibrosis research.
1. Introduction
Silicosis is a chronic occupational disease caused by exposure to dust particles containing a high level of free silica-containing silicon dioxide (SiO2), and these particles are often found in mining, quarry, metal foundries and other sandy industrial environments (Verma et al., 2008; Steenland and Ward, 2014). Silicosis is a severe problem in developing and developed countries. Although preventive efforts have been implemented for many years, silicosis remains an incurable, fatal and disabling pulmonary disease characterized by pulmonary interstitial fibrosis and silicotic nodule formation (Leung et al., 2012). Silicosis progression is due to a lack of hygienic measures and inadequate surveillance (Tse et al., 2007), and silicosis has a complex molecular and biological mechanism (Kawasaki, 2015). Additionally, the prevalence and frequency of silicosis remain very high, and currently, no effective therapies are available (Liu et al., 2015).
Despite comprehensive studies examining the toxicity of crystalline silica over the last several years, the exact etiology and particular mechanism of silicosis remain unknown. The deposition of silica particles in the alveoli of the lungs is the beginning of the pathophysiological process of silicosis. Silica particles ingested by lung cells induce an inflammatory response that produces large amounts of collagen and stimulates fibroblast overproliferation (Pollard, 2016). The principal characteristics of pulmonary fibrosis in silicosis include fibroblasts, myofibroblast proliferation, alveolar damage and deposition of extra-cellular matrix (ECM) components, which involve the abnormal remodeling of lung structure and an irreversible decline in lung function (Araya and Nishimura, 2010). Alveolar cells are the basic and essential immune barrier against a pathogenic organism and try to overcome invasion by this pathogenic organism and innate pulmonary immunity against environmental pollutants. When the free pathogenic bacteria and sand particles in the air reach the alveoli of the lung, they are triggered and removed by alveolar cells. Pulmonary fibroblasts pathologically indicate tissue fibrosis, fibroblast and myofibroblast proliferation with elevated expression levels of alpha-smooth muscle actin (α-SMA, ACTA1), collagen I (Col I, COL1A1) and collagen III (Col III, COL3A1) and deposition of ECM components (Willis et al., 2005; Tan- jore et al., 2009).
Mounting evidence has suggested that CCCH-type zinc finger proteins, such as ZC3H12A/MCPIP1, play an essential role in regulating cell proliferation and migration (Zhu et al., 2015), modulating signaling pathways and targeting mRNA degradation (Matsushita et al., 2009; Jura et al., 2012; Yang et al., 2015). ZC3H4 is a newly found member of the CCCH-type zinc finger protein ZC3H12A (Yang et al., 2015), which has been shown to be involved in inflammation and fibrosis induced by SiO2 via autophagy and endoplasmic reticulum (ER) stress (Yang et al., 2018; Jiang et al., 2019). The ER plays a significant role during the modification of posttranslational transmembrane and secretory proteins. Usually, proteins are modified in the ER before being transferred and folded. This process is energy-dependent and sensitive to ion variations, such as calcium levels, oxygen balance, and glucose supply. (Schro ̈der and Kaufman, 2005). Specific signaling pathways are activated due to ER stress, and this process is known as the unfolded protein response (UPR). Three essential proteins are involved in these signaling pathways, including inositol-requiring enzyme 1 (IRE1), activating transcription factor 6 (ATF6) and PKR-like ER kinase (PERK) (Boyce et al., 2005). ZC3H4-associated ER stress in pulmonary fibroblasts has received little consideration. In the current study, we focused on the role of ZC3H4 in pulmonary fibroblast activation via ER stress with the involvement of the MAP kinase pathway in silicosis, which will facilitate further discoveries in the progression of pulmonary fibrosis, and silicosis patients may provide a treatment strategy.
…
3. Results
3.1. SiO increases pulmonary fibroblast activation 2
Pulmonary fibroblasts pathologically indicate tissue fibrosis, fibroblast and myofibroblast proliferation with elevated expression levels of ACTA, COL1A1, and COL3A1 and deposition of ECM components (Willis et al., 2005; Tanjore et al., 2009). Fibroblast activation markers, such as COL1A1, COL3A1 and ACTA1, were measured by an immunoblotting assay (western blot), CCK-8 cell counting kit and immunofluorescence staining after SiO2 exposure (50 μg/cm2). Based on earlier experimental dosages, 50 μg/cm2 was selected for all the related experiments (Brown et al., 2007; Hao et al., 2013; Fazzi et al., 2014; Wang et al., 2015; Liu et al., 2016b). Western blotting results confirmed that fibroblast activation markers such as COL1A1, COL3A1 and ACTA1 were elevated in the presence of SiO2 after 3 h (Fig. 1A–C). In addition, the cell viability of pulmonary fibroblasts increased after 12 h of addition of silica (50 μg/ cm2) (Fig. 1D). Immunocytochemistry assays confirmed the upregulation of COL1A1 and ACTA1 (Fig. 1E and F) induced by SiO2 in pulmonary fibroblasts.
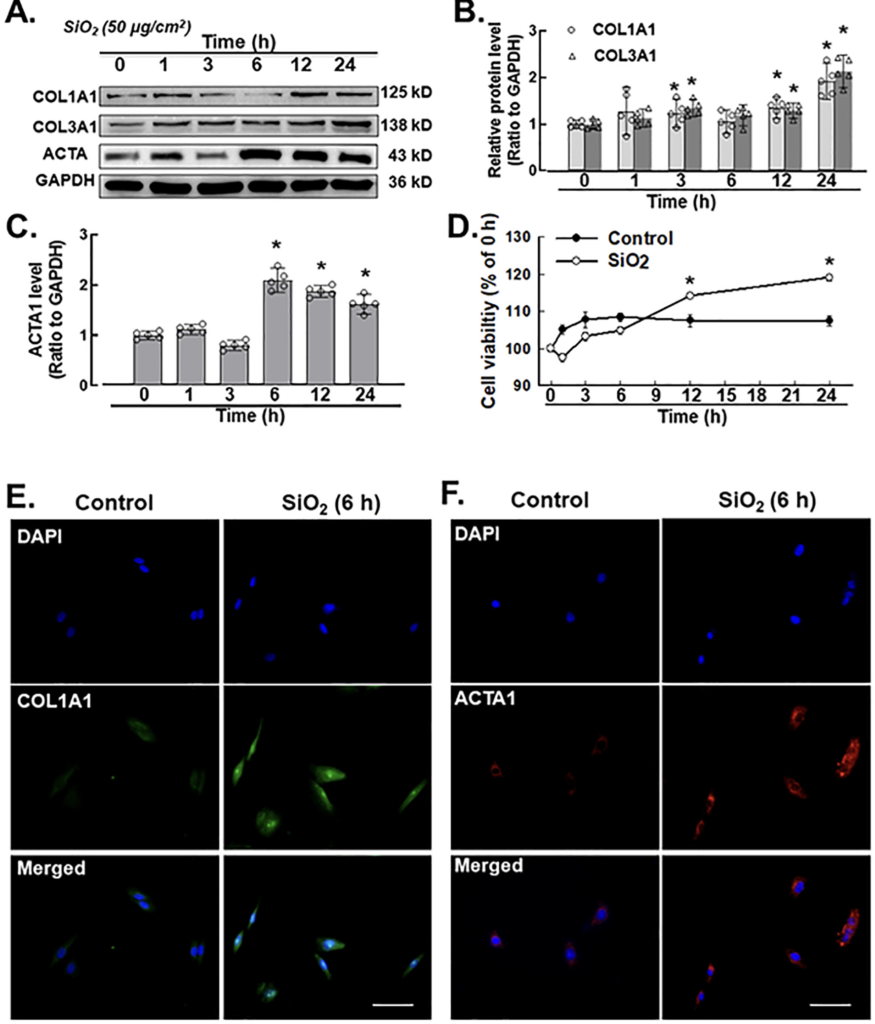
A. Demonstrative western blotting assay showing that SiO2 increased the expression level of the fibroblast inducers COL1A1 (COL I), COL3A1 (Col III) and ACTA1 (α-SMA) at different time points (hrs) in MLg cells. B. Densitometric analyses of COL1A1 (COL I) and COL3A1 (Col III) from five independent experiments at different time points (hrs) in MLg cells (n = 5): *p < 0.05 vs. the control group. C. Densitometric analyses of ACTA1 (α-SMA) from five independent experiments at different time points (hr) in MLg cells (n = 5): *p < 0.05 vs. the control group. D. CCK-8 assay showing that SiO2 increased fibroblast and myofibroblast activation in MLg cells (n = 5); *p < 0.05 vs. the control group at the corresponding time point. E. Demonstrative immu- nocytochemistry assay representing the effect of SiO2 that increased the expression level of COL1A1 (COL I) in MLg cells, scale bar = 50 μm. F. Demonstrative immunocytochemistry assay representing the effect of SiO2 that increased the expression level of fibroblast inducer ACTA1 (α-SMA) in MLg cells, scale bar = 50 μm.
3.2. SiO2 increases migration in pulmonary fibroblasts
Two-dimensional (2D) and 3D nested collagen matrix assays significantly represent the cell migration ability in in vitro culture (Grinnell, 2003; Pampaloni et al., 2007; Rhee, 2009). Compared with usual scratch assays, the nested collagen matrix (3D) assay is a reliable, quick, quantitative and easy method for determining fibroblast migration and motility in 3D models (Grinnell, 2003; Zhou and Petroll, 2010; Silva et al., 2012; Liu et al., 2015). Here, we provide new insights into the novel roles that pulmonary fibroblasts exposed to SiO2 were assessed via 2-dimensional and 3-dimensional nested collagen matrix gel assays, and their responses were examined in both models at the indicated time points. Although SiO2 induced fibroblast migration and proliferation differently in 2-dimensional and 3-dimensional nested collagen matrix gel systems, the peak response of fibroblasts was observed at 24 h. The scratch assay/wound healing assay (Fig. 2A–B) demonstrated that SiO2 significantly induced migration in MLg cells. Therefore, the schematic diagram of the 3D nested collagen matrix gel assay represents the migration of pulmonary fibroblasts after SiO2 exposure (Fig. 2C). Pulmonary fibroblasts exhibited an increase in both migration distance (Fig. 2D–E) and the number of migrated cells (Fig. 2D and F).
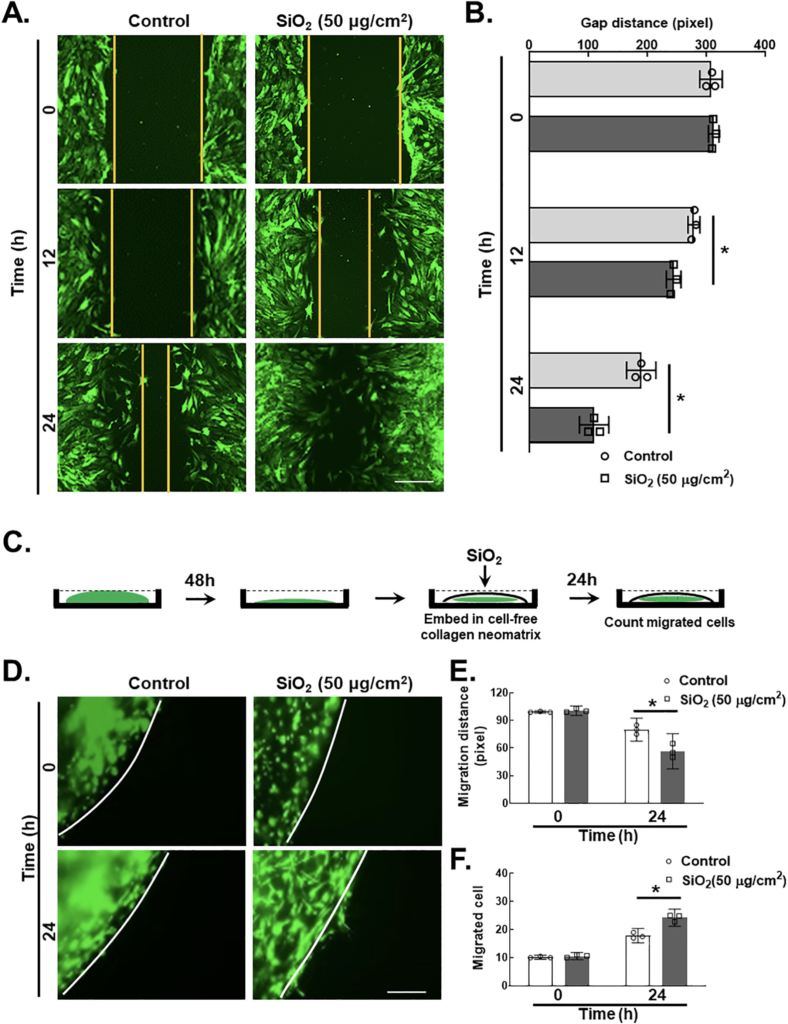
A. The 2-dimensional assay represents migratory distance after SiO2 exposure by using a scratch in pulmonary fibroblasts from 3 independent experiments. Scale bar = 100 μm. B. Quantification of gap distance from 5 independent experiments. *p < 0.05 vs. the control group at the corresponding time point after SiO2 exposure. C. The schematic diagram of the 3-dimensional nested collagen matrix gel assay represents the migration of pulmonary fibroblasts after SiO2 exposure. D. The demonstrative diagram of the 3D nested collagen matrix assay represents the induced migratory characteristic of pulmonary fibroblasts after SiO2 exposure. Scale bar = 100 μm. E. Quantification of the MLg cells was determined by using the migratory cell distance (pixel), *p < 0.05 vs. the control group F. Quantification of the MLg cells was determined by using the migrated cells, *p < 0.05 vs. the control group.
3.3. SiO2 induces ZC3H4 in pulmonary fibroblasts
A previous study from our laboratory suggested that ZC3H4 is involved in the inflammatory stage of silicosis. To establish whether ZC3H4 is also involved in late fibrosis, MLg cells were exposed to SiO2. Western blot assays confirmed that the ZC3H4 expression level was suddenly increased after SiO2 exposure in pulmonary fibroblasts at different time points (0, 1, 3, 6, 12 and 24 h) with a two-phase increase pattern (Fig. 3A–B). Immunocytochemistry confirmed the upregulation of ZC3H4 expression induced by SiO2 in pulmonary fibroblasts (Fig. 3C).
To simplify the role of ZC3H4 in fibroblast activation induced by SiO2, we transfected MLg cells with ZC3H4 NIC plasmids to specifically knockdown ZC3H4. Western blot assays confirmed that the specific knockdown of ZC3H4 in pulmonary fibroblasts significantly inhibited the upregulation of ZC3H4 with or without SiO2 (Fig. 3D–E). In the cell viability assay, ZC3H4 knockdown attenuated the increase in cell viability induced by SiO2 (Fig. 3F), indicating that ZC3H4 mediated the proliferation induced by SiO2 in fibroblasts. Moreover, ZC3H4 NIC plasmids abolished the effect of SiO2 on the migration ability of MLg cells (Fig. 3G–H). Furthermore, the COL1A1 and ACTA1 expression levels induced by SiO2 were assessed after the specific knockdown of ZC3H4 (Fig. 3I and J), in which all upregulated markers were inhibited, confirming the role of ZC3H4 in pulmonary fibroblast activation and migration.
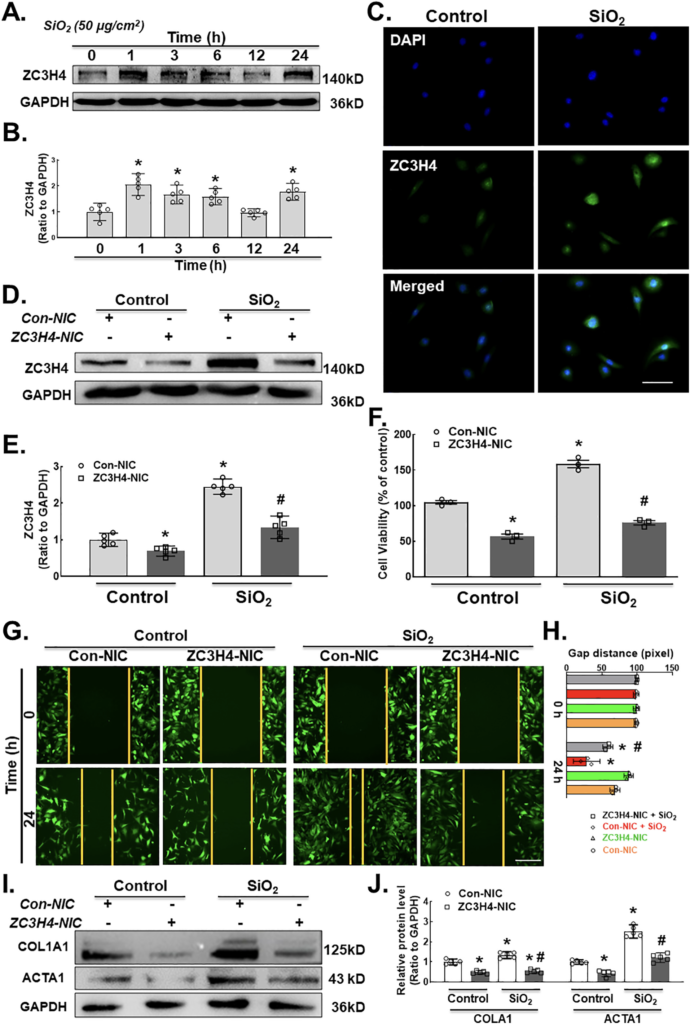
A. Demonstrative western blot assay showing the effect of SiO2 on the ZC3H4 protein with elevated expression levels at different time points (hrs) in MLg cells. B. Densitometric analyses of the ZC3H4 protein from five independent experiments in MLg cells at different time points, *p < 0.05 vs. 0 h. C. Descriptive immunocytochemistry assay showing that SiO2 increased the expression of the ZC3H4 protein in MLg cells. Scale bar = 50 μm. D. Descriptive western blot assay representing the effects of SiO2 after the transfection of pulmonary fibroblast cells with (ZC3H4-NIC) ZC3H4 double nickase plasmid to observe the downregulation of ZC3H4 proteins. E. Densitometric analyses of five independent experiments showing that SiO2 induced the expression level of ZC3H4 with attenuated CRISPR/Cas9-mediated ZC3H4 silencing in MLg cells (n = 5), *p < 0.05 vs. the Con-NIC of the control group, #p < 0.05 vs. the Con-NIC of the SiO2 group. F. CCK-8 assay showing the effects of SiO2 after the transfection of pulmonary fibroblast cells with a ZC3H4 double nickase plasmid (ZC3H4-NIC) to observe the downregulation of ZC3H4, but SiO2 induced increased fibroblast and myofibroblast cell viability in MLg cells, *p < 0.05 vs. the Con-NIC of the control group, #p < 0.05 vs. the Con-NIC of the SiO2 group. G. Descriptive pictures demonstrating that GFP-labeled MLg cells induced cell migration in a 2D assay after SiO2 exposure, but ZC3H4NIC silencing abolished pulmonary fibroblast migration. Scale bar =100 μm. H. Quantification of the gap (scratch) width was detected in a 2D nested matrix assay from five independent experiments. *p < 0.05 vs. the Con-NIC of the control group, #p < 0.05 vs. the ZC3H4-NIC of the control group. at the conforming time points. The effect of CRISPR/Cas9-mediated ZC3H4NIC silencing abolished pulmonary fibroblast migration in a 2D nested matrix assay. *p < 0.05 vs. the Con-NIC of the control group, #p < 0.05 vs. the Con-NIC of the SiO2 group. I. Demonstrative western blotting representing the effects of CRISPR/Cas9-mediated ZC3H4NIC on pulmonary fibroblasts revealed that SiO2 increased the fibroblast activation inducers COL1A1 and ACTA1. J. Densitometric analyses of COL1A1 and ACTA1 expression levels from five independent experiments in MLg cells. *p < 0.05 vs. the Con-NIC of the control group, #p < 0.05 vs. the Con-NIC of the SiO2 group.
3.4. SiO2 induces ER stress in pulmonary fibroblasts
To depict the downstream molecular mechanism of ZC3H4 on fibroblast activation, ER stress was evaluated. The ER is a key factor for cellular activities (protein modification, folding, synthesis and transport) (Lenna and Trojanowska, 2012; Sano and Reed, 2013; Urra et al., 2013). Various pathological and physiological environmental factors could affect ER homeostasis, finally causing ER stress (Tabas and Ron, 2011; Walter and Ron, 2011). ER stress is responsible for pulmonary tract infections (Zhong et al., 2011) and various types of lung disease (Wang et al., 2017; Zhang et al., 2017). UPRs are essential factors for maintaining homeostasis and initiating the BIP and CHOP pathways (Cao et al., 2017). In a western blot assay, we measured the ER stress markers ERN1 (inositol-requiring enzyme 1, IRE1α), BiP (binding immunoglobulin protein, BIP) and DDIT3 (C/EBP homologous protein, CHOP) in pulmonary fibroblasts after SiO2 exposure. MLg cells treated with SiO2 showed upregulation of ERN1, DDIT3 and BiP expression in a time-dependent manner with an ERN1 peak of 3 and 6 h, DDIT3 peak of 3 h and BiP peak of 12 h (Fig. 4A–D). Immunocytochemistry assays were applied to confirm the colocalization of ZC3H4, BiP and DDIT3 (Fig. 4E–F). To further confirm the role of ZC3H4 in ER stress, ZC3H4 was specifically knocked down. As shown in Fig. 4G–H, ZC3H4 knock-down abolished the induction of ER stress-induced by SiO2. Collectively, these results indicated that ZC3H4 is responsible for fibroblast activation induced by SiO2 via the ER stress pathway.
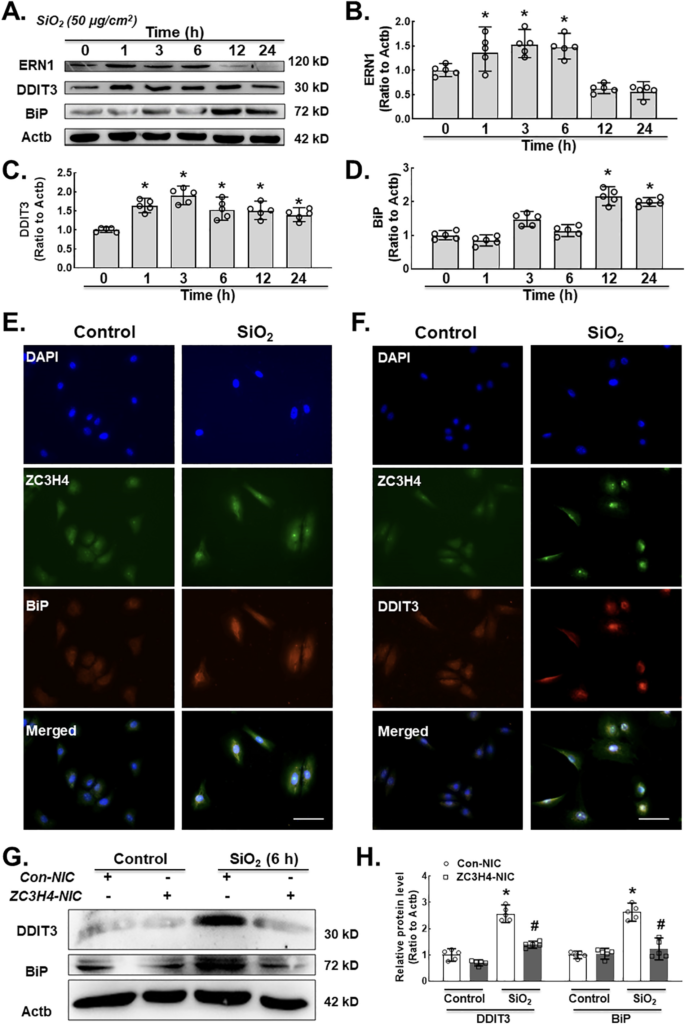
A. Demonstrative western blotting assay representing the effects of SiO2 that increased the expression level of ER stress markers such as ERN1, DDIT3 and BiP in MLg cells. B. Densitometric analyses of ERN1 from five independent experiments indicated that SiO2 induced the expression level of ERN1 in MLg cells in a time-dependent manner, *p < 0.05 vs. 0 h. C. Densitometric analyses of DDIT3 from five independent ex- periments indicated that SiO2 induced DDIT3 expression in MLg cells in a time-dependent manner, *p < 0.05 vs. 0 h. D. Densitometric analyses of BiP from five independent experiments indicated that SiO2 induced the expression level of BiP in MLg cells in a time-dependent manner, *p < 0.05 vs. 0 h. E. Demonstrative immunocytochemistry assay images demonstrating the effect of SiO2 that increased the expression levels of ZC3H4 and BiP in MLg cells, scale bar = 50 μm. F. Demonstrative immunocytochemistry assay images demonstrating the effect of SiO2 that increased the expression levels of ZC3H4 and DDIT3 in MLg cells, scale bar =50 μm. G. Demonstrative western blotting assay representing the effects of CRISPR/Cas9-mediated ZC3H4NIC on pulmonary fibroblast cells verified that SiO2 increased ER stress, such as DDIT3 and BiP. H. Densitometric analyses of DDIT3 and BiP expression levels from five independent experiments in MLg cells. *p < 0.05 vs. the Con-NIC of the control group, #p < 0.05 vs. the Con-NIC of the SiO2 group.
3.5. ZC3H4 induced ER stress via Sigmar1 in pulmonary fibroblasts
Having established the fundamental role of ZC3H4 in pulmonary fibroblast activation via ER stress in response to silica, the detailed molecular mechanism of activation of ER stress was further investigated. A previous study from our laboratory suggested that Sigmar1, a subclass of the sigma receptor family, mediated ER stress in silicosis (Cao et al., 2017). Sigmar1 is expressed in the ER with two steroid-binding domains and two transmembrane segments (Palmer et al., 2007). The molecular action of Sigmar1 was previously discovered to be a ligand-regulated receptor chaperone via ER stress (Hayashi and Su, 2007). In particular, whether ZC3H4 has a vital role in the activation of pulmonary fibroblasts via the sigmar1/ER stress pathway deserves investigation. To determine whether Sigmar1 is involved in silicosis, MLgs were exposed to SiO2 to assess the Sigmar1 level. Western blotting results confirmed that the expression level of Sigmar1 was increased in the presence of SiO2 (Fig. 5A–B), which was confirmed by immunocytochemical staining (Fig. 5C). Furthermore, the Sigmar1 expression level was assessed after the specific knockdown of ZC3H4, in which ZC3H4- NIC suppressed sigmar1 upregulation after SiO2 exposure (Fig. 5D–E). To further understand the regulatory effect of ZC3H4 on Sigmar1, Co-IP was conducted, in which there was a direct interaction between ZC3H4 and Sigmar1 (Fig. 5F). Taken together, these results verified that ZC3H4 and Sigmar1 are involved in the pulmonary fibroblast activation induced by SiO2.
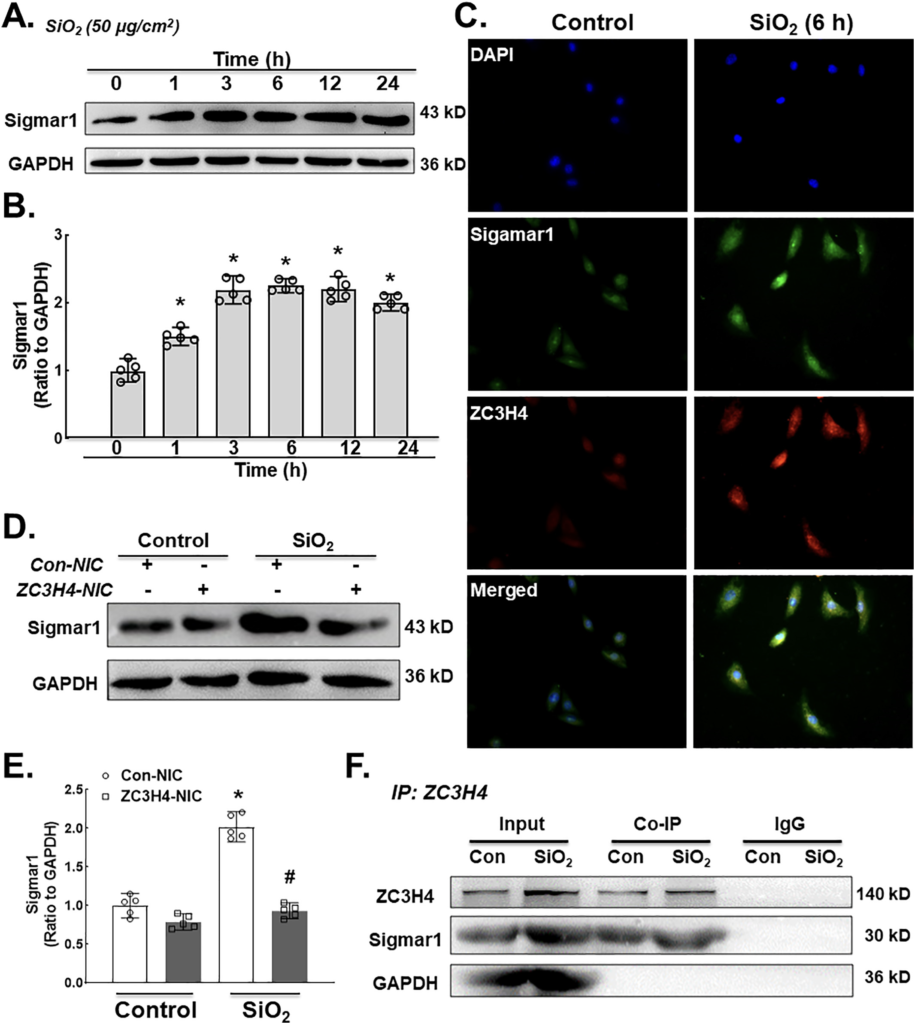
A. Demonstrative western blotting assay showing the effect of SiO2 that increased the protein expression level of Sigmar1 in a time-dependent manner in MLg cells. B. Densitometric analyses represented the Sigmar1 protein from five independent experiments in MLg cells in a time-dependent manner, *p < 0.05 vs. 0 h. C. Descriptive immunocytochemistry assay showing that SiO2 increased the expression of Sigmar1 and ZC3H4 protein in MLg cells. D. Demonstrative western blotting assay representing ZC3H4NIC on pulmonary fibroblast cells that verified that SiO2 increased the Sigmar1 protein. E. Densitometric analyses of Sigmar1 protein expression levels from five independent experiments in MLg cells. *p < 0.05 vs. the Con- NIC of the control group, #p < 0.05 vs. the Con-NIC of the SiO2 group. F. To clarify that the interaction between ZC3H4 and Sigmar1 was assessed after SiO2 exposure, we examined the immunoprecipitation of ZC3H4 in whole-cell lysates followed by immunoblotting for ZC3H4.
3.6. MAPK and PI3K pathways are involved in SiO2-induced ZC3H4 upregulation
To further clarify the possible regulatory mechanism of ZC3H4, the involvement of the MAPK and P13K/Akt pathways was investigated since the MAPK and P13K/Akt pathways play an essential role in cell proliferation, migration and activation (Yao et al., 2009, 2010). First, the short-term effect of SiO2 on ZC3H4 was measured, in which a rapid and transient increase in ZC3H4 was observed. Then, the MAPK signaling pathway was assessed. As shown in Fig. 6C–D, phosphorylation of Mapk1, Mapk8 and Mapk14 was increased after SiO2 exposure. Moreover, Akt phosphorylation showed a slight but significant increase after SiO2 exposure (Fig. 6E–F). To assess the role of the MAPK or PI3K pathway in the regulation of ZC3H4, specific pharmacological inhibitors were applied. MLg cells were pretreated with a Mapk1 (SP600125) in- inhibitor, Mapk8 (SB203580) inhibitor, Mapk14 (U0126) inhibitor and P13K (LY294002) inhibitor separately for 1 h, after which SiO2 was applied. As shown in Fig. 6G–H, all inhibitors attenuated the increase in ZC3H4 and sigmar1 induced by SiO2, confirming the role of the MAPK and PI3K pathways in the regulation of ZC3H4 and Sigmar1. Further- more, fibroblast activation markers such as COL1A1 and ACTA1, as well as the ER stress markers DDIT3 and BiP, were inhibited by pretreatment with inhibitors (Supplementary Fig. 4A-E). These results demonstrated that the MAPK and PI3K/Akt pathways play a significant role in the regulation of ZC3H4.
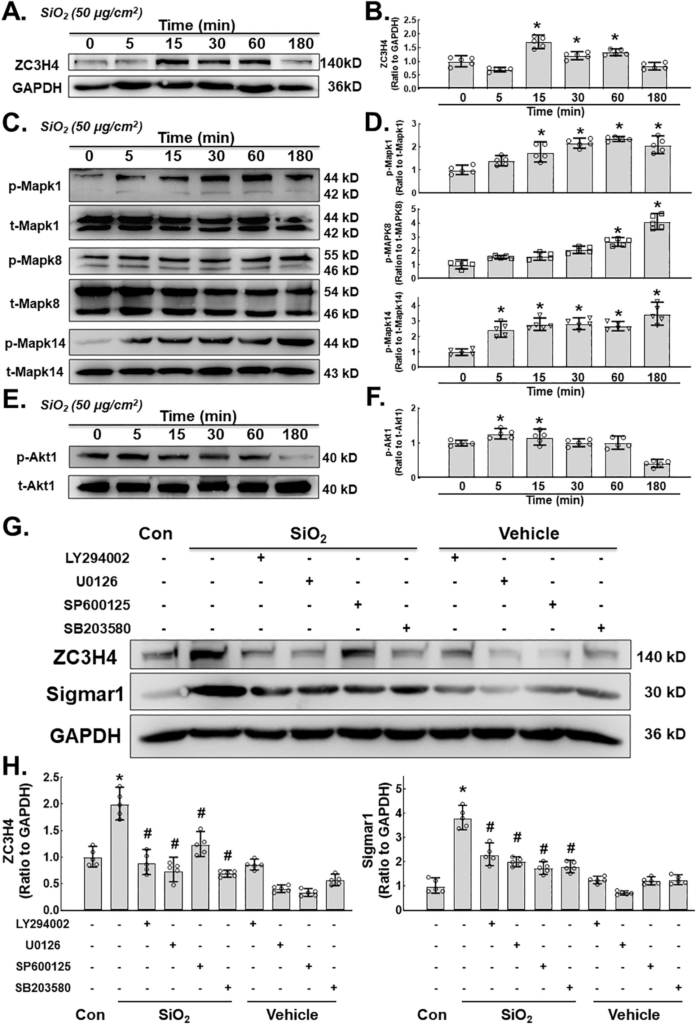
A. Demonstrative immunoblotting assay demonstrating the effects of SiO2 that increased the expression levels of ZC3H4 in MLg cells in a time-dependent manner. B. Densitometric analyses of ZC3H4 representing the effects of SiO2 that increased the expression levels from five independent experiments in MLg cells in a time-dependent manner, *p < 0.05 vs. 0 min. C. Demonstrative western blotting assay showing that Mapk1, Mapk8 and Mapk14 were elevated after SiO2 exposure in MLg cells in a time (min)-dependent manner. D. Densitometric analyses of Mapk1, Mapk8, and Mapk14 representative the effects of SiO2 that increased the expression levels from five independent experiments in MLg cells in a time-dependent manner, *p < 0.05 vs. 0 min. E. Demonstrative western blotting assay showing that P13K/Akt was elevated after SiO2 exposure in MLg cells in a time (min)-dependent manner. F. Densitometric analyses of P13K/Akt representative the effects of SiO2 that increased the expression levels from five independent experiments in MLg cells in a time-dependent manner, *p < 0.05 vs. 0 min. G. Demonstrative immunoblotting assay demonstrating the effects of SiO2 that increased the expression levels of ZC3H4 and Sigmar1 were attenuated by pretreatment with MAPK (Mapk1, Mapk8, Mapk14) and P13K/Akt phosphorylation pathway inhibitors. H. Densitometric analyses representing the effects of SiO2 that increased the expression levels of Sigmar1 and ZC3H4 (F) were attenuated by pretreatment with MAPK (Mapk1, Mapk8, Mapk14) and P13K/Akt phosphorylation pathway inhibitors, n = 5, *p < 0.05 vs. the control group, #p < 0.05 vs. the SiO2 group.
3.7. Induction of ZC3H4 by SiO2 promotes a further increase in ZC3H4
Interestingly, SiO2 induced a two-phase increase pattern in ZC3H4 (Fig. 3A–B), in which the early increase was due to the activation of MAPK and PI3K. The mechanism of the late increase in ZC3H4 deserves to be investigated since pulmonary fibrosis is a chronic pathological process. As ZC3H4-induced ER stress may increase the UPR in the ER lumen (Boyce and Yuan, 2006), whether ER stress affects the abnormal increase in ZC3H4 needs to be clarified. To our surprise, while salubrinal inhibited the increase in ER stress markers (Supplementary Fig. 5A–B), salubrinal reversed the SiO2 effect in the upregulation of ZC3H4 in pulmonary fibroblasts (Fig. 7A–B). Furthermore, tunicamycin, a specific ER stress inducer, was applied to further explore the role of ER stress. As expected, tunicamycin induced the expression of ER stress markers in a dose-dependent manner, and ZC3H4 was also upregulated (Fig. 7C–F). Furthermore, pulmonary fibroblasts were further exposed to tunicamycin, in which the ZC3H4 level was increased at a peak at 24 h, while there was no rapid increase within 6 h, which showed a different pattern compared with direct SiO2 exposure (Fig. 7G–H). Immunocytochemistry assays confirmed the upregulation of ZC3H4 expression after 50 μM tunicamycin in pulmonary fibroblasts (Fig. 7I). These results demon- strated that ER stress was involved in the upregulation and activation of ZC3H4 in pulmonary fibroblasts in the late phase, indicating that the positive feedback loop (PFL) may be involved (Fig 8). Taken together, these results demonstrate that ZC3H4/ER stress plays a significant role in the activation of pulmonary fibroblasts.
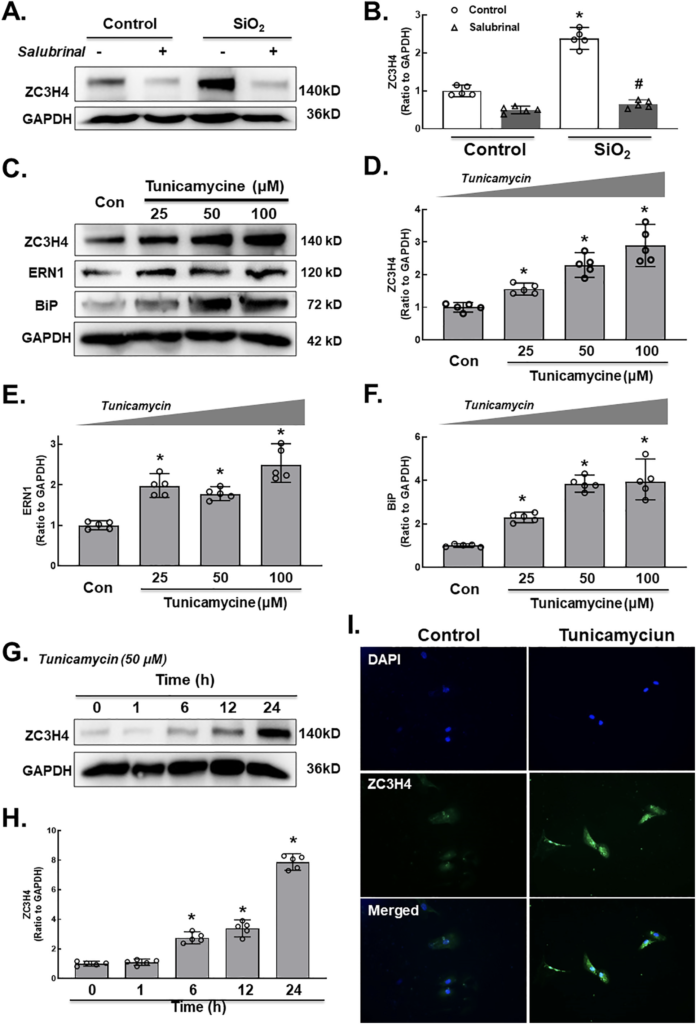
A. Demonstrative western blotting assays showed that salubrinal was applied to MLg cells, indicating that ER stress was involved in the upregulation of ZC3H4. B. Densitometric analyses of ZC3H4, n = 5, *p < 0.05 vs. the control group, #p < 0.05 vs. the SiO2 group. C. Demonstrative western blotting assays showed that tunicamycin was applied to MLg cells, indicating that ER stress was involved in the upre- gulation of ZC3H4. D. Densitometric analyses of five independent experiments indicated the effects of tunicamycin on ZC3H4 in MLg cells. *p < 0.05 vs. the control group. Densitometric analyses of five independent experiments indicated the effects of tunicamycin on ERN1 (E) and BiP (F) in MLg cells. *p < 0.05 vs. the control group. G. Demonstrative western blotting assays showed that tunicamycin induced the upregulation of ZC3H4 in a time-dependent manner. H. Densitometric an- alyses of five independent experiments indicated the effects of tunicamycin on ZC3H4 in MLg cells. *p < 0.05 vs. the 0 h group. I. Descriptive immunocytochemistry assay representing the effect of tunicamycin responsible for the elevated expression level of ZC3H4 in MLg cells.
4. Discussion
Pulmonary fibrosis is a sequela of many pulmonary diseases, such as idiopathic pulmonary fibrosis and pneumoconiosis. Pulmonary fibroblasts pathologically indicate tissue fibrosis and fibroblast and myofibroblast proliferation (Willis et al., 2005; Tanjore et al., 2009). The current study principally focused on the role of ZC3H4 and sigmar1 in the proliferation, migration and activation of pulmonary fibroblasts exposed to SiO2, which may provide a target for pulmonary fibrosis treatment, and the detailed molecular mechanism was also investigated.
Pulmonary fibroblasts clinically represent the proliferation of fibroblasts and myofibroblasts with the deposition of ECM components (Willis et al., 2005; Tanjore et al., 2009). The zinc finger protein family has been shown to play a potential role in the inflammatory response and represents a downregulation of the inflammatory reaction, as well as fibroblast activation, such as ZC3H12A/MCPIP1 (Jura et al., 2012; Yang et al., 2015). ZC3H4 is similar to ZC3H12A, and both belong to the same family of CCCH-type zinc finger proteins (Yang et al., 2015). Interestingly, ZC3H12A may act as a molecular signal in the initiating pathway or as a functional factor to induce or inhibit inflammation, mainly depending on the time frame. The current study suggested a similar pattern of ZC3H4, in which ZC3H4 initiated ER stress as a signaling molecule in the early stage and promoted fibroblast migration and proliferation in the late stage, forming PFLs. PFLs are responsible for producing all-or-none responses by continuously converting graded in- puts into discrete outputs (Ferrell Jr, 2002; Mitrophanov and Groisman, 2008) and transforming fibroblasts to myofibroblasts (Elson et al., 2019). Mounting evidence has shown that fibrosis in pneumoconiosis and IPF is a chronic pathological process in which both signaling molecules and the TGF-β pathway are involved. CCCH-type zinc finger proteins participate in PFLs to initiate fibrosis and maintain the fibrosis process, indicating a suitable target for blocking or reserving this process. This two-phase increase pattern of ZC3H4 may guide new strategies for the treatment of fibrosis by an early blockade of ZC3H4 initiation.
Although the comprehensive mechanism of ZC3H4 in ER stress must be clarified, the present study recommends a novel role of ZC3H4 in ER stress. Previous studies have shown that ZC3H4 promotes epithelial-to-mesenchymal transition (EMT) via ER stress, which participates in the fibrosis induced by SiO2 (Jiang et al., 2019). Currently, the detailed mechanism by which ZC3H4 induces ER stress was investigated, in which ZC3H4 directly interacts with Sigmar1, which is expressed in the ER and a subclass of the sigma receptor family (Palmer et al., 2007). The molecular action of Sigmar1 was previously discovered to be a ligand- regulated receptor chaperone via ER stress. Sigmar1 has been shown to be involved in fibroblast activation (Cao et al., 2017). Interestingly, the mRNA levels of both ZC3H4 and Sigmar1 did not show a significant change due to SiO2 exposure, while these two proteins have been shown to be regulated by noncoding RNAs, such as circular RNAs (circRNAs) (Cao et al., 2017; Yang et al., 2018). circRNAs are involved in cell proliferation and transformation, and their misregulation is related to diseases and can have phenotypes in animal models (Chen, 2020). However, given the low abundance of most circRNAs, whether the translation of certain circRNAs is crucial for their endogenous function remains unclear. Hence, the direct interaction between ZC3H4 and sigmar1 was investigated in the current study, but it did not rule out the role of circRNA in the regulation of these two proteins. On the other hand, the current study was carried out in a mouse fibroblast cell line, since a previous study from our lab showed a similar response of mouse and human fibroblasts to SiO2 (Liu et al., 2016a; Zhou et al., 2018; Chu et al., 2019), in which SiO2 promoted activation in both mouse and human fibroblasts. However, recent studies have shown that both mouse (Balb/c 3 T3 and HEKn) and human (HDF) fibroblast cell lines were sensitive to the phototoxic potential of chlorpromazine (CPZ), while HDF showed insensitivity to phototoxic evaluation (Rai et al., 2011), indicating the inconsistency of cell response to different stimulation in the different species cell lines. Whether SiO2 promotes a similar pattern of ZC3H4 in both mouse and human fibroblasts deserves further inves- tigation. Taken together, these results verified that ZC3H4 and Sigmar1 were involved in the pulmonary fibroblast activation induced by SiO2.
Although the present study still focused on the activation of pulmonary fibroblasts, it delivered a new direction to treat silicosis. MAPK signaling pathways are responsible for the activation of transcription factors. Activation of MAPK is involved in the upstream signaling pathways for ER stress induction (Choi et al., 2011). In addition, MAPK pathways such as p38, JNK, and ERK were also stimulated by ER stress inducers and played a significant role in different cell signaling pathways and cell death (Wang and Ron, 1996; Urano et al., 2000; Arai et al., 2004; Yang et al., 2007). Inositol-requiring enzyme 1 (IRE1) is responsible for the activation and stimulation of MAPK (Urano et al., 2000). After stress, fibroblast cells inhibit migration due to abnormal signaling pathway responses, although ER stress is responsible for lung diseases (Hsu et al., 2017; Wang et al., 2017; Zhang et al., 2017). Due to the rapid and transient increase in ZC3H4 after SiO2 exposure within 6 h, MAPK initiated the expression of ZC3H4. Recently, Ets-like protein-1 (Elk-1) has been shown to mediate ZC3H4 expression (Jiang et al., 2020) in epithelial cells, while Elk-1 also induces ZC3H12A in macrophages (Kasza et al., 2010), which indicates a similar upstream regulatory mechanism for the CCCH zinc finger protein family. Taken together, these results demonstrate that MAPK/ZC3H4/ER stress plays a significant role in the activation of pulmonary fibroblasts.
5. Conclusion
Our study revealed a link between ZC3H4 and pulmonary fibroblast activation via the sigmar1/ER stress pathway in silicosis; SiO2 exposure enhanced the MAPK and P13K/Akt phosphorylation pathways with the involvement of ZC3H4. Interestingly, ZC3H4 has been shown to induce ER stress with the participation of a PFL, providing a novel therapeutic strategy for treating silicosis (Fig. 8).
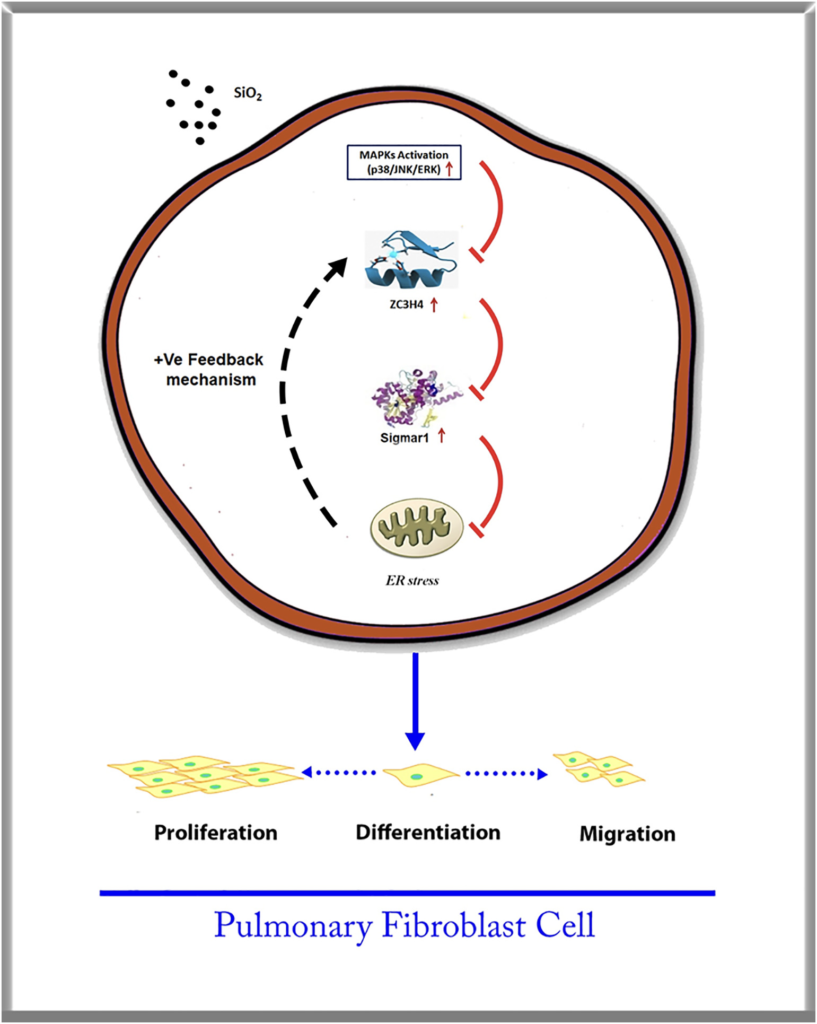
ZC3H4 promotes pulmonary fibroblast acti- vation with the involvement of MAPK (Mapk1, Mapk8 and Mapk14), and P13K/ Akt phosphorylation pathways via sigmar1/ ER stress in silicosis play a significant role in fibroblast proliferation, migration and dif- ferentiation. The interaction between ZC3H4 and Sigmar1 promotes the ER stress pathway with a PFL that enhances the pro- liferation, migration and differentiation abilities with the increased synthesis of collagen, which may lead to fibrosis induced by SiO2.