Roles of Transmembrane Protein 97 (TMEM97) in Adipose Tissue and Skeletal Muscle
By Masafumi Tenta, Jun Eguchi, and Jun Wada
Excerpt from the article published in Acta Medica Okayama, 2022 Jun; 76 (3):235-245. DOI: 10.18926/AMO/63717
Editor’s Highlights
- Sarcopenic obesity, the combination of sarcopenia and obesity, is defined as reduced skeletal muscle mass and muscle strength in parallel with increased adiposity.
- Adipose tissue is the major site for the storage of excess energy from food intake as triacyl-glycerol (TAG).
- Adipose tissue is now recognized as a master regulator of the body’s energy balance and nutritional homeostasis.
- Transmembrane protein 97 (TMEM97), also known as Sigma-2 receptor (σ2R) or meningioma-associated protein (MAC30), is a member of the family of insulin-like growth factor binding proteins, and it regulates cholesterol transport.
- TMEM97 acts on metabolic physiology in at least two organs, adipose tissue and skeletal muscle.
- Overexpressing TMEM97 in adipose tissue and in skeletal muscle showed induced systemic insulin resistance. And a global deletion of TMEM97 protects against diet-induced obesity and insulin resistance.
- TMEM97 appears to play a critical role in metabolic function and in the development of sarcopenic obesity.
Abstract
The combination of sarcopenia and obesity (sarcopenic obesity) is associated with the development of metabolic syndrome and cardiovascular events. The molecular pathways that develop sarcopenic obesity have studied intensively. Transmembrane protein 97 (TMEM97) is 176 amino acids conserved integral membrane protein with four transmembrane domains that is expressed in several types of cancer. Its physiological significance in adipose tissue and skeletal muscle has been unclear. We studied TMEM97-transgenic mice and mice lacking TMEM97, and our findings indicate that TMEM97 expression is regulated in adipose tissue and skeletal muscle from obesity. TMEM97 represses adipogenesis and promotes myogenesis in vitro. Fat-specific TMEM97 transgenic mice showed systemic insulin resistance. Mice overexpressing TMEM97 in skeletal muscle exhibited systemic insulin resistance. Mice lacking TMEM97 were protected against diet-induced obesity and insulin resistance. These phenotypes are associated with the effects of TMEM97 on inflammation genes in adipose tissue and skeletal muscle. Our findings indicate that there is a link between TMEM97 and chronic inflammation in obesity.
Introduction
Adipose tissue and skeletal muscle are important organs involved in insulin sensitivity and body weight regulation. White adipose tissue is the major site for the storage of excess energy from food intake as triacyl-glycerol (TAG). TAG is broken down into free fatty acids and glycerol, and it is released to be supplied to other tissues. Recent research has demonstrated that adipose tissue secretes a number of bioactive proteins and regulates systemic insulin sensitivity and energy metabolism. Adipose tissue is now recognized as a master regulator of the body’s energy balance and nutritional homeostasis.
Obesity is defined as the expansion of white adipose tissue. A positive imbalance between energy intake and energy expenditure results in the development of obesity. Adipose tissue dysfunction is one of the causes of obesity-induced insulin resistance. Adipose tissue dysfunction in obesity is characterized by adipocyte hypertrophy (a pro-inflammatory adipose tissue phenotype) and fibrosis, which result in ectopic fat deposition and inflammation in visceral adipose tissue [1, 2]. Visceral adipose tissue and skeletal muscle are known to interact and share common inflammatory pathways in obesity. Ectopic fat deposition in skeletal muscle enhances systemic insulin resistance and metabolic abnormalities [3, 4]. It is thus important to identify the mechanisms involved in adipose tissue dysfunction and impaired skeletal muscle function.
Transmembrane protein 97 (TMEM97), also known as meningioma-associated protein (MAC30), encodes a conserved integral membrane protein of 176 amino acids with four transmembrane domains. TMEM97 is a member of the family of insulin-like growth factor binding proteins, and it regulates cholesterol transport [5-7]. Tmem97 mRNA is expressed in the fetal liver and plays an important role in liver development and differentiation [8]. It was recently reported that TMEM97 is highly expressed in several types of cancers (breast, oesophagus, stomach, and colon cancers) and can act as a tumor suppressor or oncogene in carcinoma [9, 10]. The sigma-2 receptor is known to be an 18-to 21-kDa membrane protein and highly expressed in the liver, kidney and the central nervous system as well as in several cancers. Recently the sigma-2 receptor was identified as TMEM97 using mass spectrometry from liver samples [11]. However, there is no report about the roles of TMEM97 in adipose tissue and skeletal muscle of obesity-induced insulin resistance.
To investigate the functional roles of TMEM97 in adipose tissue and skeletal muscle, we studied TMEM97-transgenic mice and mice lacking TMEM97. Our results indicate that (i) TMEM97 is expressed in mouse fat and muscle and is developmentally regulated, and (ii) TMEM97 plays a significant role in adipogenesis and myogenesis. We also observed that mice lacking TMEM97 display increased systemic insulin sensitivity.
…
Results
TMEM97 is expressed in mouse adipose tissue and skeletal muscle and is regulated during differentiation.
To the best of our knowledge, there are no published reports of TMEM97 expression or function in adipose tissue and skeletal muscle. The present study’s QPCR results demonstrated that Tmem97 mRNA is expressed in murine white adipose tissue (WAT) and skeletal muscle (Fig. 1A). The expression of Tmem97 mRNA showed developmental regulation during adipogenesis and myogenesis in cultured adipocytes and myocytes. Tmem97 mRNA was expressed in pre-adipocytes and was then repressed after the induction of differentiation (Fig. 1B). C2C12 cells are an immortalized mouse myoblast cell line, and they differentiate into myotubes under serum starvation. The expression of Tmem97 mRNA was induced during the differentiation of C2C12 cells (Fig. 1C).
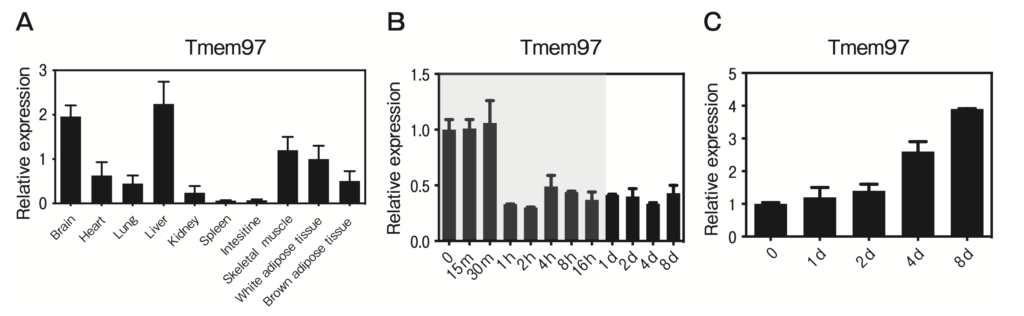
TMEM97 is expressed in adipose tissue and skeletal muscle and is regulated during adipogenesis and myogenesis.
A, The QPCR-based expression of TMEM97 in tissues from male C57BL/6J mice. Data are the mean±SEM fold induction relative to Tmem97 mRNA expression in white adipose tissue (WAT); all values are normalized to 36B4 mRNA expression. n=5; B, TMEM97 expression during the differentiation of 3T3-L1 cells. Note that the time scale is not linear; the shaded area indicates time points <24 hr. Data are normalized to the 36B4 mRNA expression and are expressed as the fold induction relative to Tmem97 mRNA on day 0. n=3; C, TMEM97 expression during C2C12 differentiation. Data are normalized to 36B4 mRNA expression and are expressed as the fold induction relative to the Tmem97 mRNA expression on day 0. n=3.
TMEM97 expression was induced in WAT and skeletal muscle from obese mice.
To investigate the roles of TMEM97 in adipose tissue and skeletal muscle from obese mice, we investigated whether TMEM97 expression is regulated in DIO mice and ob/ob mice. The expression of Tmem97 mRNA was induced in epididymal fat from DIO and ob/ob mice (Fig. 2A), and the expression of Tmem97 mRNA was induced in skeletal muscle from ob/ob mice (Fig. 2B).
Adipose tissue contains adipocytes, immune cells, and vascular cells. To examine which cell type(s) express TMEM97, we fractioned epididymal fat into adipocytes and the stromal vascular fraction (SVF), and we performed a QPCR to measure the Tmem97 mRNA expression. Tmem97 mRNA was predominantly expressed in the SVF of the epididymal adipose tissue (Fig. 2C). Interestingly, Tmem97 mRNA levels were induced in the mature adipocyte fraction from ob/ob mice (Fig. 2D). However, Tmem97 mRNA expression was not induced in the SVF from ob/ob mice (Fig. 2E). These data suggest that TMEM97 plays a role in mature adipocytes of obese mice.
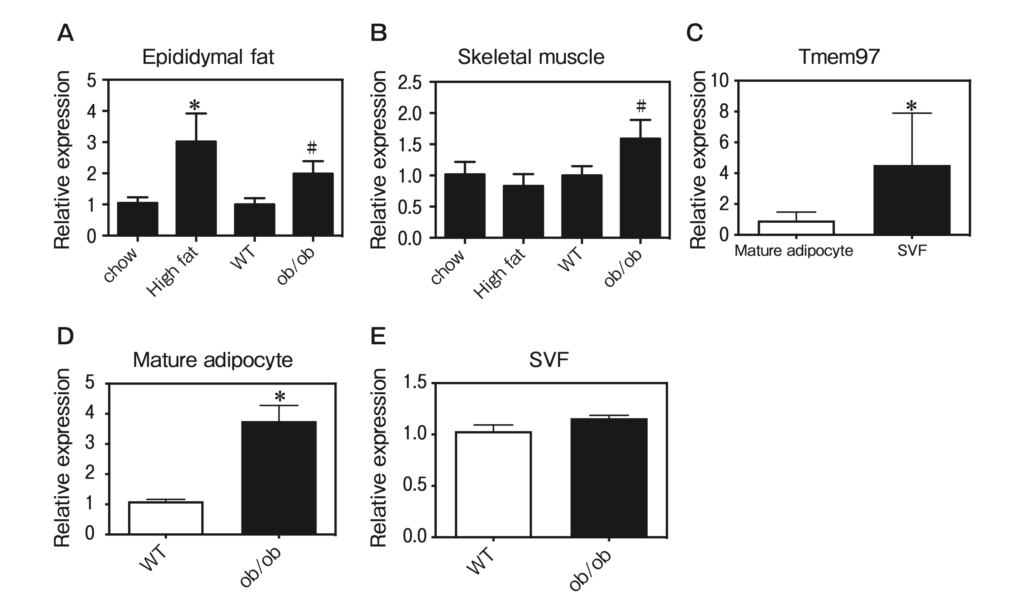
TMEM97 expression was induced in obese mice.
A, Tmem97 mRNA expression in WAT from obese mice. Tmem97 mRNA expression was measured by a QPCR in epididymal fat from mice fed a high-fat diet and ob/ob mice. Data are normalized to the 36B4 mRNA expression and are expressed as the fold induction relative to the mice fed a standard chow diet (*p<0.05) or wild-type (WT) mice (#p<0.05). Results are mean±SD. n=5; B, Tmem97 mRNA expression in skeletal muscle from obese mice. Tmem97 mRNA expression was measured by a QPCR in skeletal muscle from mice fed a high-fat diet and ob/ob mice. Data are normalized to the 36B4 mRNA expression and are expressed as the fold induction relative to the mice fed a standard chow diet (*p<0.05) or WT mice (#p<0.05). Results are mean±SD. n=5; C, Tmem97 mRNA expression was assessed in samples of fractioned fat pads from C57BL/6J mice. SVF: stromal-vascular fraction. Results are the mean±SD fold induction relative to the Tmem97 mRNA expression in the SVF. n=5. *p<0.05 vs. mature adipocytes; D, Tmem97 mRNA expression in mature adipocytes from ob/ob mice. Results are the mean±SD fold induction relative to Tmem97 mRNA expression in mature adipocytes from WT mice. n=5. *p<0.05 vs. mature adipocytes from WT mice; E, Tmem97 mRNA expression in the SVF from ob/ob mice. Results are the mean±SD fold induction relative to the Tmem97 mRNA expression in the SVF from WT mice. n=5. *p<0.05 vs. the SVF from WT mice.
TMEM97 regulates adipogenesis and myogenesis.
We sought to determine whether TMEM97 plays a functional role in adipogenesis. TMEM97 was introduced by lentiviral transduction into 3T3-L1 preadipocytes, which were then treated with dexamethasone, methylisobutylxanthine, and insulin. The overexpression of TMEM97 resulted in the inhibitions of adipogenesis and adipocyte gene expression (Fig. 3A, B). We next asked whether the overexpression of TMEM97 affects myogenesis. TMEM97 was introduced by lentiviral transduction into C2C12 cells, the differentiation of which was induced by replacing 10% FBS with 2% HS. The overexpression of TMEM97 resulted in enhanced myogenesis and enhanced myocyte gene expression on day 0 (Fig. 3C, D). These data suggest a specific role of TMEM97 in adipogenesis and myogenesis.
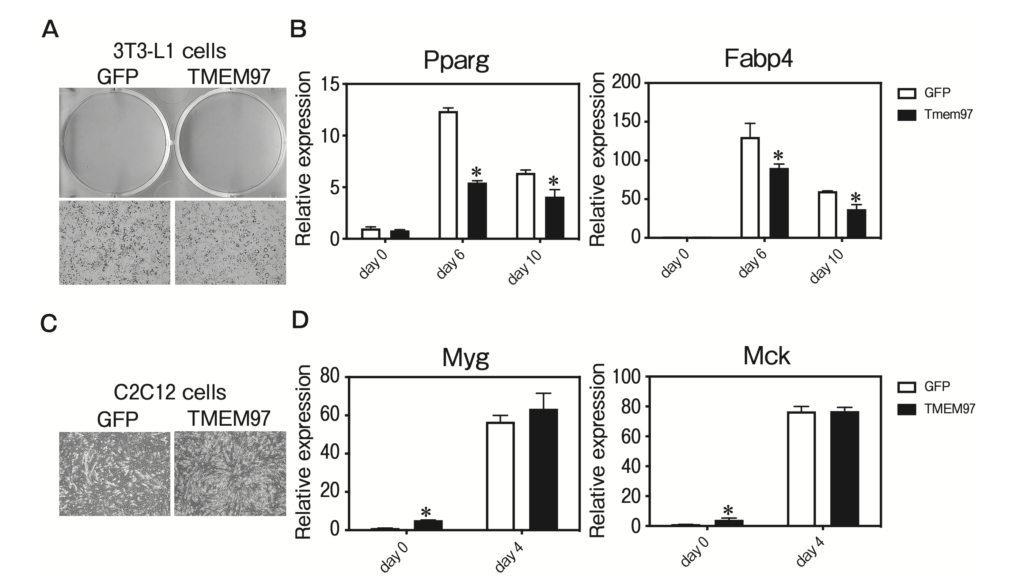
TMEM97 represses adipogenesis and enhances myogenesis.
A, 3T3-L1 pre-adipocytes were transduced with lentivirus expressing TMEM97 or GFP, and differentiated with dexamethasone, methylisobutylxanthine, and insulin (DMI). Cells were stained with oil red O at 10 days post-DMI treatment; B, Cells shown in panel A were harvested at days 0, 6, and 10 post-DMI treatment, and a QPCR was used to determine the levels of Pparg and Fabp4 genes relative to mRNA expression on day 0. n=3; C, C2C12 cells were transduced with lentivirus expressing TMEM97 and differentiated by replacing 10% fetal bovine serum (FBS) with 2% horse serum (HS). Cells were observed 4 days after replacing 10% FBS with 2% HS; D, Cells shown in panel C were harvested at day 0 and day 4 after replacing 10% FBS with 2% HS, and a QPCR was used to determine the levels of Myg and Mck genes relative to mRNA expression on day 0. n = 3.
Fat-specific overexpression of TMEM97 results in systemic insulin resistance.
We next sought to extend our in vitro findings to a more physiologically relevant system. We generated aP2-TMEM97 transgenic mice by using a transgene consisting of the murine Tmem97 gene driven by the aP2 promoter, which is highly expressed in adipose tissue (Fig. 4A). The Tmem97 mRNA expression in epididymal fat from the aP2-TMEM97 transgenic mice was significantly increased compared to that in the wild-type (WT) mice (Fig. 4B). The total body weights of the aP2-TMEM97 transgenic mice were not significantly different from those of the WT mice maintained on the standard chow diet (Fig. 4C). However, the aP2-TMEM97 transgenic mice demonstrated significantly impaired insulin tolerance (Fig. 4D). The QPCR analysis revealed that genes related to inflammation were increased in the WAT from aP2-TMEM97 transgenic mice (Fig. 4E). These data suggest that TMEM97 overexpression induced adipose tissue inflammation in the aP2-TMEM97 transgenic mice.
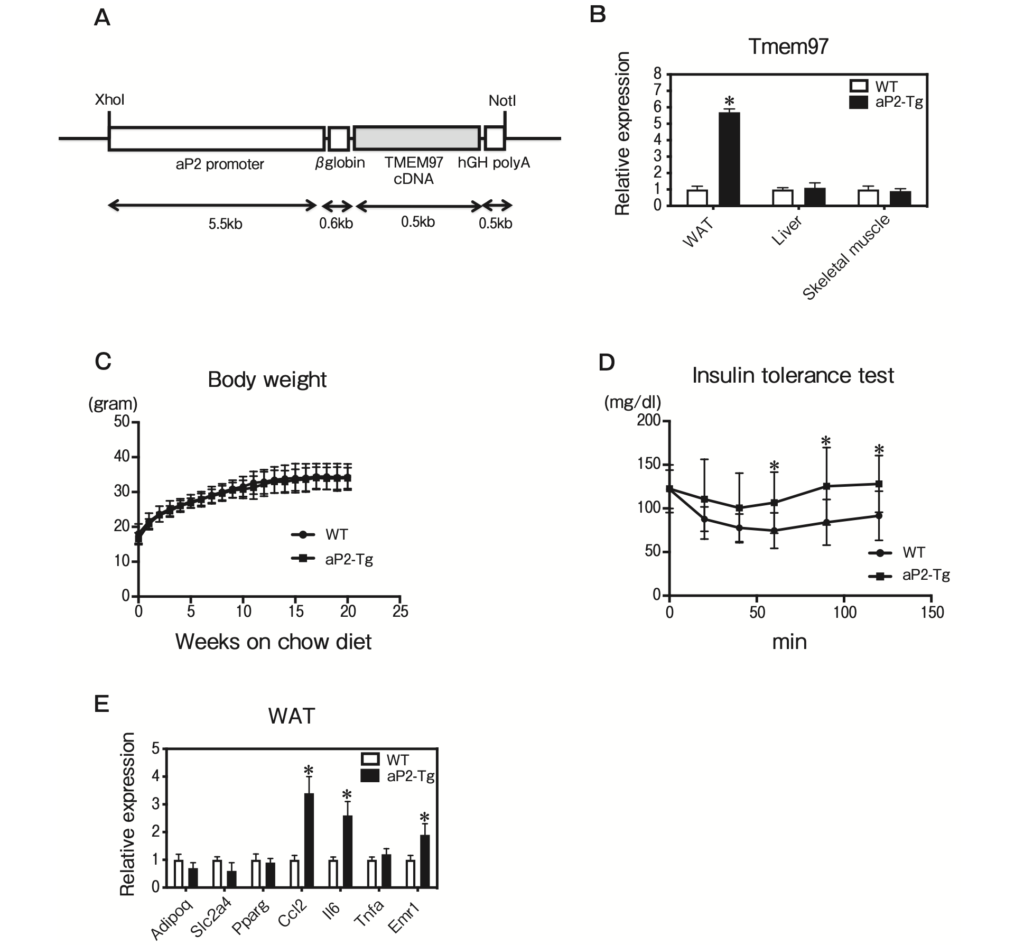
Characterization of fat-specific TMEM97 transgenic mice.
A, Structure of the aP2-TMEM97 expression vector. The DNA fragments contained aP2 promoter, rabbit β-globin intron, coding region of TMEM97 cDNA, and hGH polyadenylation site (polyA); B, Tmem97 mRNA expression in epididymal WAT, liver, and skeletal muscle of male WT and aP2-TMEM97 transgenic (aP2-Tg) mice (n = 3). Data are normalized to 36B4. *p<0.05 relative to WT mice. Results are mean±SD; C, Body weights of the male WT and aP2-Tg mice maintained on the standard chow diet (n=10). Results are mean±SD; D, Insulin tolerance test. Blood glucose in 20-week-old WT and aP2-Tg mice fed the standard chow diet was determined at the indicated times after an intraperitoneal injection of a bolus of insulin. (n=10). Results are mean±SD; E, QPCR analysis of the indicated mRNAs in epididymal WAT from 12-week-old WT and aP2-Tg mice maintained on the standard chow diet (n = 5) under fasting condition. All samples are normalized to 36B4. Results are mean ± SD. *p<0.05 relative to the WT mice.
Skeletal muscle-specific overexpression of TMEM97 results in systemic insulin resistance.
To investigate the effect of TMEM97 on skeletal muscle function, we generated HSA-TMEM97 transgenic mice by using a transgene consisting of the murine Tmem97 gene driven by human α-skeletal actin, which is predominantly expressed in skeletal muscle (Fig. 5A). The Tmem97 mRNA expression in skeletal muscle from the HSA-TMEM97 transgenic mice was significantly increased compared to that of the WT mice (Fig. 5B). The total body weights of the HSA-TMEM97 transgenic mice were not significantly different from those of the WT mice maintained on the high-fat diet (Fig. 5C). However, the HSA-TMEM97 transgenic mice demonstrated significantly impaired insulin tolerance (Fig. 5D). SDH staining and COX staining detect slow twitch (type I) fibers, which are mitochondria-rich and oxidative [13]. Our histological analysis of skeletal muscle revealed that the SDH and COX staining was significantly decreased in HSA-TMEM97 transgenic mice (Fig. 5E). The QPCR analysis demonstrated that genes related to inflammation were increased in the skeletal muscle of HSA-TMEM97 transgenic mice (Fig. 5F). These data suggest that TMEM97 overexpression reduced mitochondrial function and induced inflammation in skeletal muscle from the HSA-TMEM97 transgenic mice.
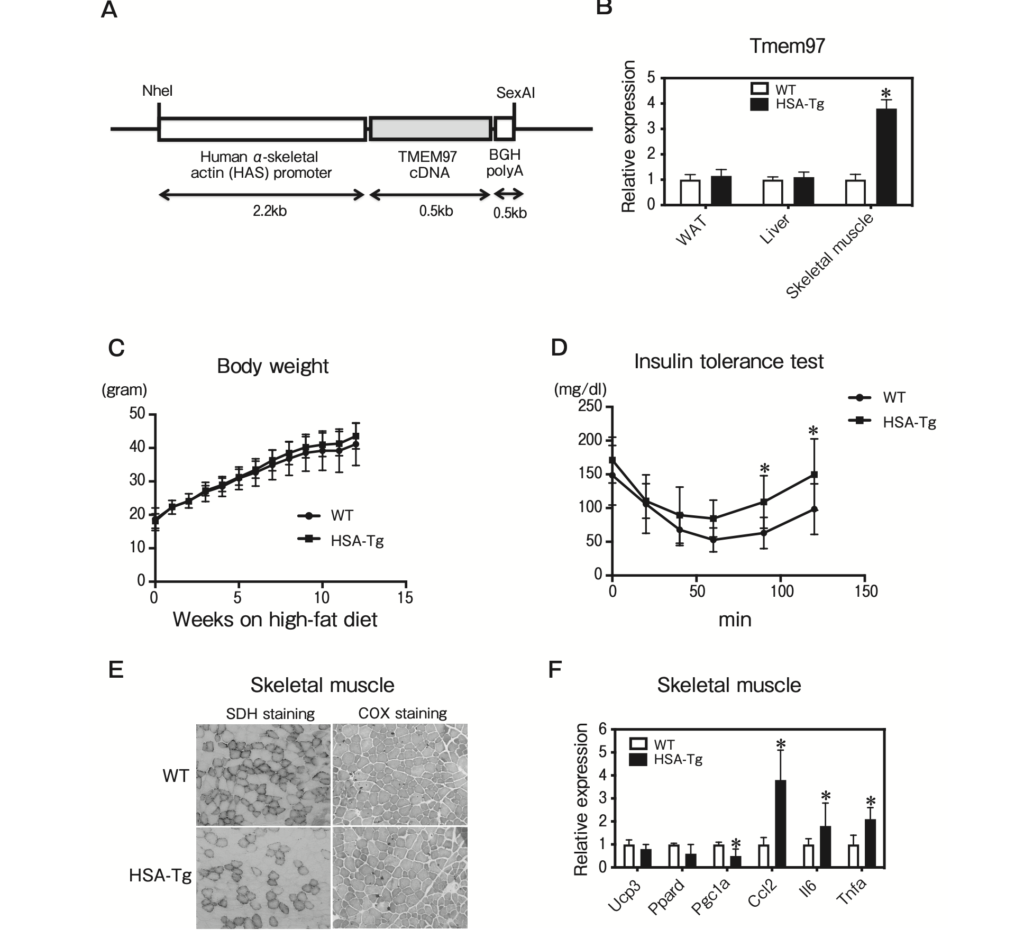
Characterization of skeletal muscle-specific TMEM97 transgenic mice.
A, Structure of the human α-skeletal actin (HSA)- TMEM97 expression vector. The DNA fragments contained HSA promoter, coding region of TMEM97 cDNA, and BGH polyadenylation site (polyA); B, Tmem97 mRNA expression in epididymal WAT, liver, and skeletal muscle of male WT and HSA-TMEM97 transgenic (HSA-Tg) mice (n=3). Data are normalized to 36B4. *p<0.05 relative to the WT mice. Results are mean±SD; C, Body weights of male WT and HSA-Tg mice on the standard chow diet (n=10). Results are mean±SD; D, Insulin tolerance test. Blood glucose in 20-week-old WT and HSA-Tg mice fed the standard chow diet was determined at the indicated times after an intraperitoneal injection with a bolus of insulin (n = 10). Results are mean ± SD. *p < 0.05; E, SDH and COX staining of frozen skeletal muscle sections from 12-week-old mice; F, QPCR analysis of the indicated mRNAs in skeletal muscle from 12-week-old WT and HSA-Tg mice on the chow diet (n=5) under fasting condition. All samples are normalized to 36B4. Results are mean±SD. *p<0.05 relative to the WT mice.
Global deletion of TMEM97 resulted in improved systemic insulin resistance in DIO mice.
TMEM97 KO mice were modestly protected against diet-induced obesity. The body weights of the TMEM97 KO mice were significantly lighter lower than those of the WT mice maintained on a high-fat diet (Fig. 6A). The epididymal fat and liver weights in the TMEM97 KO mice were not significantly different from those of the WT mice maintained on a high-fat diet (Fig. 6B).
Interestingly, the TMEM7 KO mice showed improved glucose and insulin tolerance after 12 weeks of high-fat feeding (Fig. 6C, D). DIO induced inflammation and adipocyte dysfunction in adipose tissue. Given that TMEM97 plays a pro-inflammatory role in adipose tissue and skeletal muscle, its deletion improved DIO-induced inflammation in adipose tissue and skeletal muscle. The results of the QPCR analysis revealed that the mRNA expressions of Adipoq, Slc2a4, and Pparg were increased and the mRNA expressions of Ccl2, Il6, Tnfa, and Emr1 were decreased in the epididymal fat from TMEM97 KO mice (Fig. 6F). There was no detectable difference in hepatic lipid content between the WT and TMEM97 KO mice (Fig. 6E). In skeletal muscle, the histological analysis showed that the SDH and COX staining was significantly increased in the TMEM97 KO mice (Fig. 6E). Consistent with the histology analysis, the QPCR results revealed that the mRNA expressions of Ucp3, Ppard, and Pgc1a were increased and the mRNA expressions of Ccl2 and Tnfa were decreased in the skeletal muscle from TMEM97 KO mice (Fig. 6G). Taken together, these results suggest that global TMEM97 deficiency is sufficient for the improvement of DIO-induced insulin resistance through the improvement of mitochondrial function and the suppression of inflammation in adipose tissue and skeletal muscle.
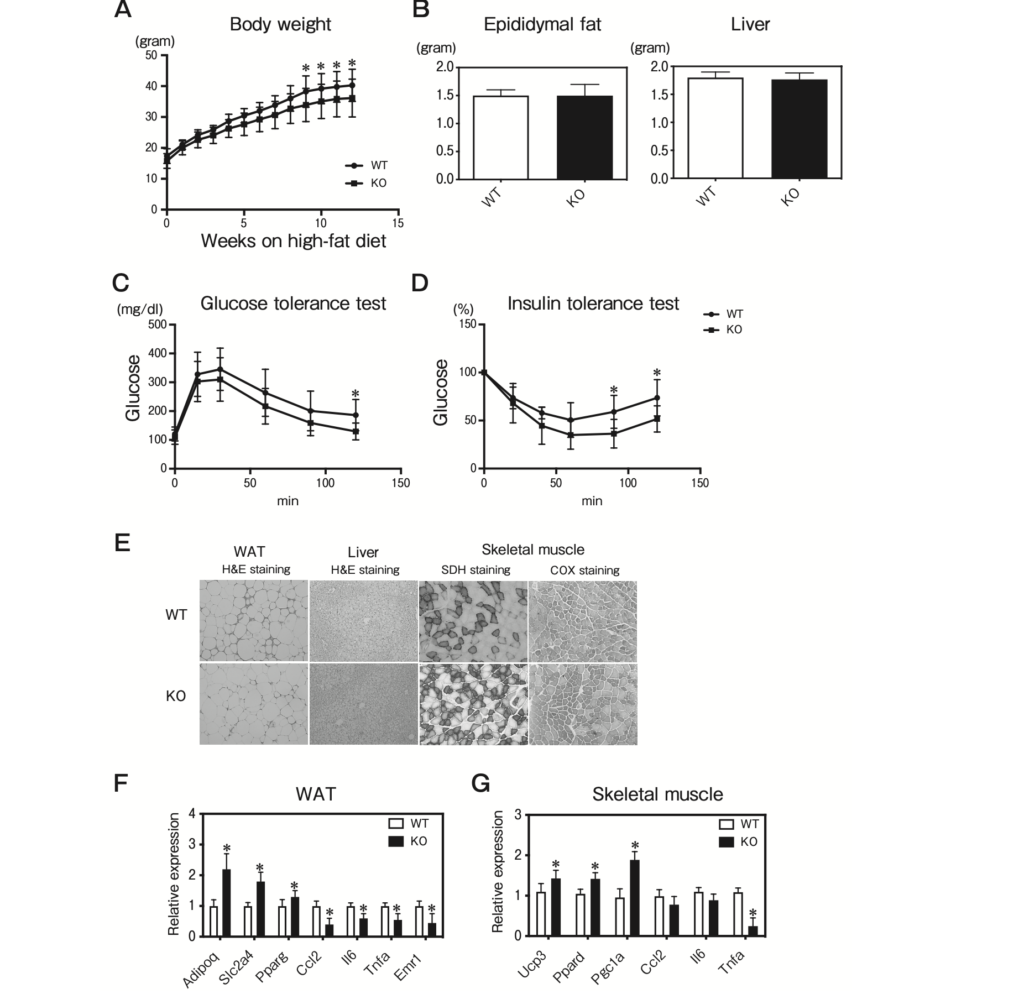
Characterization of TMEM97 knockout mice.
A, Body weights of male WT and Tmem97 knockout (KO) mice maintained on a high-fat diet (n=10). Results are mean±SD. *p<0.05; B, Tissue weights of male WT and Tmem97 KO mice on the high-fat diet (n=8). Results are mean±SD; C, Glucose tolerance test. Blood glucose in 16-week-old WT and KO mice fed the high-fat diet was determined at the indicated times after intraperitoneal injection with a bolus of glucose (n=10). Results are mean±SD. *p<0.05; D, Insulin tolerance test. Blood glucose in 18-week-old WT and KO mice fed the high-fat diet was determined at the indicated times after an intraperitoneal injection with a bolus of insulin. (n=10). Results are mean±SD. *p<0.05; E, Hematoxylin and eosin staining of epididymal WAT and liver from 12-week-old mice. SDH and COX staining of frozen skeletal muscle sections from 12-week-old mice; F, QPCR analysis of the indicated mRNAs in epididymal WAT and skeletal muscle from 12-week-old WT and KO mice on the high-fat diet (n=5) under fasting condition. All samples are normalized to 36B4. Results are mean±SD. *p<0.05 relative to the WT mice.
Discussion
We have been interested in the metabolic role of TMEM97, and our present findings demonstrated that TMEM97 is expressed in adipose tissue and skeletal muscle and plays an important metabolic role in adipose tissue and skeletal muscle from obese mice. TMEM97 was induced in the adipose tissue and skeletal muscle by a high-fat diet, and it appears to act as an “accelerator” of inflammatory gene expression. This is suggested by our observations that (i) the overexpression of TMEM97 in fat or skeletal muscle enhanced the expressions of inflammatory genes, and (ii) the global ablation of TMEM97 had the opposite effect. Consistent with these findings, the mice with an overexpression of TMEM97 in fat or skeletal muscle were systemic insulin-resistant, and the mice with a global deletion of TMEM97 were protected against diet-induced obesity and insulin resistance.
TMEM97 was identified as a meningioma-related gene [10 , 14]. TMEM97 has been studied mainly in the context of cancer cells. Tmem97 mRNA is expressed in the human fetal liver but not in the human adult liver, suggesting that is has an important role in the growth and differentiation of human liver [8]. It has been reported that TMEM97 exhibits altered expression in several types of cancer; for example, the level of TMEM97 expression is low in most pancreatic cancer cells, whereas TMEM97 is highly expressed in several types of cancers (breast, esophagus, stomach, and colon cancers), indicating that TMEM97 acts as a tumor suppressor or oncogene in different types of carcinomas [9]. A recent study using genome-wide expression profiling in combination with targeted RNAi screening identified TMEM97 as an SREBP target gene that enriches endo-/lysosomal compartments and binds Nieman-Pick C1 protein in sterol-depleted cells. This study indicated that TMEM97 plays a role in cholesterol and lipid metabolism [6, 7].
The mechanisms by which TMEM97 regulates functional roles of adipose tissue and skeletal muscle remain unclear. Several research groups have reported that myokines and adipokines regulate metabolic pathways and inter-organ crosstalk in autocrine, paracrine, and endocrine manners [15]. One possibility is that TMEM97 is responsible for controlling effects of myokines and adipokines. Further studies are required to investigate the mechanisms underlying the functions of TMEM97 in adipose tissue and skeletal muscle.
Sarcopenic obesity, the combination of sarcopenia and obesity, is defined as reduced skeletal muscle mass and muscle strength in parallel with increased adiposity. Increased fat mass causes skeletal muscle atrophy and impairs mitochondria function in skeletal muscle. Recent studies suggested that sarcopenic obesity is associated with an increased risk of metabolic syndrome and cardiovascular events [4, 16]. There is significant interest in developing drugs as therapy for sarcopenic obesity. The molecular pathways by which sarcopenic obesity develops have been under intensive study. TMEM97 appears to play a critical role in mitochondrial function and inflammation of adipose tissue and skeletal muscle from obesity. TMEM97 may thus play key role in the development of sarcopenic obesity.
In summary, our data suggest that TMEM97 acts on metabolic physiology in at least two organs, i.e., adipose tissue and skeletal muscle. We observed that TMEM97 repressed adipogenesis in adipose tissue. Mice overexpressing TMEM97 specifically in adipose tissue showed induced systemic insulin resistance. In skeletal muscle, TMEM97 promoted myogenesis. Mice overexpressing TMEM97 specifically in skeletal muscle exhibited induced systemic insulin resistance. Mice lacking TMEM97 showed reduced diet-induced insulin resistance. TMEM97 appears to play a critical role in adipose tissue and skeletal muscle from obesity. TMEM97 may play key roles in metabolic function independent of its effects on cancer biology and cholesterol metabolism.

