ADV368-LAD for Late-onset Alzheimer’s Disease:
- First-in-class bispecific Sigma1 agonist and Sigma2 Antagonist
- Revolutionary design
- High Equipotency
- Selective Sigma1 and Sigma2
- Oral monotherapy
- Pharmaceutical profile
- Low potential for clinical drug-drug interactions
Mechanism of Action on Late-onset Alzheimer’s Disease
ADV368 has been precisely designed to combine a potent S1R agonist activity which improves neurogenesis and the survival of mature neurons, autophagy, and maintains neuronal homeostasis, with an equally potent S2R antagonist activity to restore damaged cellular processes such as cholesterol synthesis, and protein trafficking, and blocks Abeta (Aβ) oligomer binding.
In Late-onset Alzheimer’s disease Aβ accumulation may induce calcium influx in neurons and activate CaN/NFAT signaling pathway to initiate BIP expression, which recruits ubiquitin ligase E3, leading to the ubiquitination and degradation of the sigma-1 protein. ADV368 is believed to activate sigma-1 levels, protect against hypoxia‐induced damage, alleviate neuroinflammation, increase the density of dendritic spines, promote neurogenesis and neuritogenesis, combined with the displacement of Aβ oligomers, and mitigation of its synaptotoxic effects.
Preclinical studies have shown that Sigma-2 antagonist activity displaces Aβ oligomers and prevents further binding, reducing brain damage and allowing control mechanisms to be re-activated, while Sigma-1 agonist activity promotes intensive stimulation of hippocampal neurogenesis, indicated by the quantitative increase of stem cells and immature neurons, in addition, it prevents amyloid-β-induced neurotoxicity in pathways associated with endoplasmic reticulum stress, neuroinflammation, hyper-reactive astrocyte density and degree of astrogliosis. Additionally, the Sigma-1 receptor is associated with the increased brain-derived neurotrophic factor (BDNF) in the hippocampus and with BDNF secretion by brain-derived astrocytes and neuronal cells, and BDNF decreases neurotrophins in AD patient brains, can inhibit the generation of Aβ and prevent Aβ-induced neurotoxicity, and improve learning and memory abilities in AD animal models.
The sigma-1 receptor also stabilizes IP3R and enhances the ER–mitochondrial Ca2+ transfer and alleviates mitochondrial abnormalities associated with reduced MAM functionality, such as increased mitophagy. Also alleviates BBB disruption via upregulating the levels of VEGF and LRP-1 in AD.
Therefore, ADV368 represents a disease-modifying therapeutic strategy and offers the opportunity to reverse late-onset Alzheimer’s disease by reducing the accumulation of Aβ and correcting its effects.
Learn more
The revolution of combining two Sigma chaperone receptors as targets with specific functionality for each in a single compound
Sigma receptors have very complex pharmacology that is further complicated by the lack of commercial functional assays to identify and classify the σ ligands, limiting the potential use of these unique pluripotent protein chaperones as therapeutic targets.
In addition, the severity of current incurable pathologies, such as Alzheimer’s disease, has clearly demonstrated the need to develop drugs with more than one target to achieve a full therapeutic effect in complex late-stage diseases.
We have learned that molecules that attack more than one target may possess a safer profile than single-target molecules, and the concept of dual-target (bispecific) drugs has progressed rapidly and dramatically from an emerging paradigm when first enunciated in the early 2000s to one of the hottest topics in drug discovery today.
Advantx Pharmaceuticals is the only biopharmaceutical company able to develop compounds that combine Sigma receptor chaperone targets with specific functionality thanks to the clinical experience of single-target drugs, powerful quantitative systems pharmacology, a computer-based methodology that combines preclinical neuropharmacology, neurophysiology, multiple animals models, and existing clinical information, which allows the most effective combinations in an animal model to be translated to a virtual human patient.
In complex diseases, such as late-onset Alzheimer’s disease, where single-target drugs have failed or show severe limitations, bispecific drugs emerge as a new and more effective therapeutic option.
In-vivo preclinical validation in progress.
- Hippocampal Aβ injection in rats
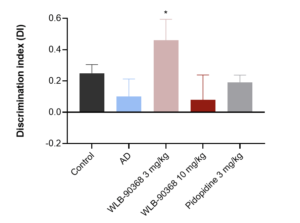
- T-Maze: ADV368 at 3 mg/kg po reverses short-term memory impairment induced by the hippocampal Aβ injection in rat
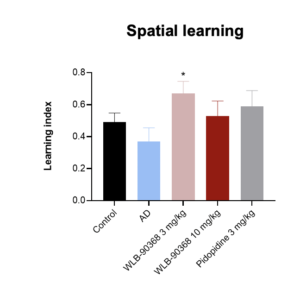
- MWM: ADV368 at 3 mg/kg po improves spatial learning and memory impairments induced by hippocampal Aβ injection in rats
- T-Maze: ADV368 at 3 mg/kg po reverses short-term memory impairment induced by the hippocampal Aβ injection in rat
